Summary
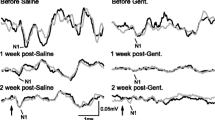
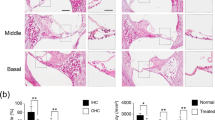
Using the experimental guinea pig model, in which total and selective destruction of the cochlear receptor revealed saccular acoustic responses, ultrastructural observations were made on otolithic organs using electron microscopy. Scanning electron microscope studies of otoconia from the saccule and utricle of experimental animals did not show any differences with control animals. Quantitative and qualitative observations of sensory neural junctions of type I cells also did not show any abnormality. These results confirm the extremely selective toxicity of amikacin for the cochlea, and thus strongly suggest that an acoustic reception by the saccule is a normal feature of the guinea pig inner ear.
Similar content being viewed by others
References
Aran JM, Cazals Y, Erre JP (1979) Conflicting electrophysiological and anatomical data from drug impaired guinea pig cochleas. Acta Otolaryngol (Stockh) 87:300–309
Bennett MLV, Nakajima Y, Pappas GD (1967) Physiology and ultrastructure of electrotonic junctions. I. Supramedullary neurons. J Neurophysiol 30:161–179
Cazals Y, Aran JM, Erre JP, Guilhaume A, Hawkins JE jr (1979) “Neural” responses to acoustic stimulation after destruction of cochlear hair cells. Arch Otorhinolaryngol 224:61–70
Cazals Y, Aran JM, Erre JP, Guilhaume A (1980) Acoustic responses after total destruction of the cochlear receptor: brainstem and auditory cortex. Science 210:83–86
Cazals Y, Aran JM, Erre JP, Guilhaume A, Aurousseau C (1983) Vestibular acoustic reception in the guinea pig: a saccular function? Acta Otolaryngol (Stockh) 95:211–217
Engstrom H, Ades HW, Hawkins JE jr (1965) The vestibular sensory cells and their innervation. Symp Biol Hung 5:21–41
Gulley RL, Bagger Sjoback D (1979) Freeze-fracture studies on the synapse between the type I hair cell and the calyceal terminal in the guinea-pig vestibular system. J Neurocytol 8:591–603
Hamilton DW (1968) The calyceal synapse of type I vestibular hair cells. J Ultrastruct Res 23:98–114
Harada Y, Sugimoto Y (1977) Metabolic disorder of otoconia after streptomycin intoxication. Acta Otolaryngol (Stockh) 84:65–71
Hawkins JE jr (1976) Drug ototoxicity. In: Keidel WD, Neff WD (eds) Handbook of sensory physiology. Springer, Berlin Heidelberg New York, pp 707–748
Hawkins JE jr, Johnsson LG, Aran JM (1969) Comparative tests of gentamicin ototoxicity. J Infect Dis 119:417–426
Johnsson LG, Hawkins JE jr (1972) Striai atrophy in clinical and experimental deafness. Laryngoscope 83:1105–1125
Johnsson LG, Wright CC, Preston RE, Henry PG (1980) Streptomycin-induced defects of the otoconial membrane. Acta Otolaryngol (Stockh) 89:401–406
Koitchev K, Guilhaume A, Cazals Y, Aran JM (1982) Spiral ganglion changes after massive aminoglycoside treatment in the guinea pig. Acta Otolaryngol (Stockh) 94:431–438
Sotelo C (1975) Morphological correlates of electronic coupling between neurons in mammalian nervous system. In: Santini M (ed) Golgi Centennial Symposium Proceedings. Raven Press, New York, pp 344–365
Spoendlin H (1966) Some morphofunctional and pathological aspects of the vestibular sensory epithelia. In: Second Symposium on the Role of Vestibular Organs in the Exploration of Space, NASA Report No. SP-115, Washington, D.C., pp 99–116
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cazals, Y., Guilhaume, A. Otoconia and neural junctions of type I hair cells in amikacin-treated guinea pigs presenting saccular acoustic responses. Arch Otorhinolaryngol 242, 155–160 (1985). https://doi.org/10.1007/BF00454415
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00454415