Abstract
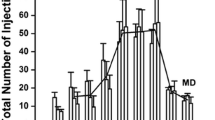
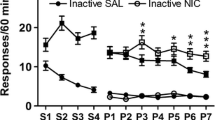
Lever-Pressing responses of 55 rats were reinforced with IV-delivered cocaine (0.2 mg/kg) or saline under conditions of continuous access for 15 24-h sessions. The rats also responded on tongue-operated drinking devices for deliveries of a 3% (w/v) glucose +0.125% (w/v) sacharin (G+S) solution or water. The effects of removing these substances on behavior maintained by G+S, water, cocaine, or saline were compared in 11 groups. Terminating cocaine access produced a decrease in G+S drinking and an increase in food and water intake. In contrast, a group of rats that did not initially self-administer G+S showed increases in G+S drinking when cocaine was removed, and G+S-maintained responding persisted when cocaine was reinstated. Substitution of water for G+S produced a nearly two-fold increase in cocaine-reinforced behavior but no change in IV-delivered saline self-administration in a control group. A group that did not initially self-administer cocaine increased its infusion rate to over 400 infusions per day as soon as G+S was replaced with water. The effect of presenting cocaine to a group that responded for G+S alone was to decrease G+S intake, but there was only a a transient decrease in water intake in the control group. Likewise, presentation of G+S to a group of rats self-administering cocaine resulted in a decrease in infusions, but saline infusions did not change in a control group. Generally, there was an increase in food and water intake during cocaine removal, but food and water intake did not vary systematically with the removal or presentation of G+S. The results suggest that behavior reinforced by IV-delivered cocaine can be substantially altered by the discontinuation or presentation of G+S, an orally self-administered nondrug reinforcer.
Similar content being viewed by others
References
Ahlenius S, Engel J (1974) Behavioral stimulation induced by ethanol withdrawal. Pharmacol Biochem Behav 2:847–850
Aigner C, Balster RL (1978) Choice behavior in rhesus monkeys: cocaine versus food. Science 201:534–535
Beardsley PM, Meisch RA (1981) A precision drinking device for rats tested with water, etonitazene, and ethanol. Pharmacol Biochem Behav 14:871–876
Beardsley PM, Balster RL, Harris LS (1985) Behavioral dependence in rhesus monkeys following chronic THC administration. In: Harris LS (ed) Problems of drug dependence. US Government Printing Office, Washington, DC, pp 111–117
Bergmann F, Lieblich I, Cohen E, Ganchrow JR (1985) Influence of intake of sweet solutions on the analgesic effect of a low dose of morphine in randomly bred rats. Behav Neural Biol 44:347–353
Branch MN, Dearing ME, Lee DM (1980) Acute and chronic effects of Δ-tetrahydrocannibinol on complex behavior of squirrel monkeys. Psychopharmacology 7:247–256
Carney JM (1982) Effects of caffeine, theophylline and theobromine on schedule controlled responding in rats. Br J Pharmacol 75:451–454
Carroll ME (1985) Concurrent phencyclidine and saccharin access: presentation of an alternative reinforcer reduced drug intake. J Exp Anal Behav 43:131–144
Carroll ME (1987a) A quantitative assessment of phencyclidine dependence produced by oral self-administration in rhesus monkeys. J Pharmacol Exp Ther 242:405–412
Carroll ME (1987b) Self-administration of orally-delivered phencyclidine and ethanol under concurrent fixed-ratio schedules in rhesus monkeys. Psychopharmacology 93:1–7
Carroll ME, Boe IN (1982) Increased intravenous drug self-administration during deprivation of other reinforcers. Pharmacol Biochem Behav 17:563–567
Carroll ME, Lac ST (1987) Cocaine withdrawal produces behavioral disruptions in rats. Life Sci 40:2183–2190
Carroll ME, France CP, Meisch RA (1979) Food deprivation increases oral and intravenous drug intake in rats. Science 205:319–321
Carroll ME, France CP, Meisch RA (1981a) Intravenous self-administration of etonitazene, cocaine and phecyclidine in rats during food deprivation and satiation. J Pharmacol Exp Ther 217:241–247
Carroll ME, Santi PA, Rudell RL (1981b) A microcomputer system for the control of behavioral experiments. Pharmacol Biochem Behav 14:415–417
Catania AC (1963) Concurrent performance: a baseline for the study of reinforcement magnitude. J Exp Anal Behav 6:299–300
de la Garza R, Bergman J, Hartel CR (1981) Food deprivation and cocaine self-administration. Pharmacol Biochem Behav 15:141–144
Driscoll JW, Lockard RB (1967) Replacement behavior following deprivation of a saccharin solution or water. Psychon Sci 7:21–22
Fischman MW (1984) The behavioral pharmacology of cocaine in humans. In: Grabowski J (ed) Cocaine: pharmacology effects and treatment of abuse. NIDA Res Monogr 50. US Government Printing Office, Washington, DC, pp 72–91
Ford RD, Balster RL (1976) Schedule-controlled behavior in the morphine dependent rat. Pharmacol Biochem Behav 4:569–573
Gawin FH, Kleber HD (1986) Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Arch Gen Psychiatry 43:107–113
Gellert VF, Sparber SB (1977) A comparison of the effects of naloxone upon body weight loss and suppression of fixed-ratio operant behavior in morphine dependent rats. J Pharmacol Exp Ther 201:44–54
Gentry RT, Dole VP (1987) Why does a sucrose choice reduce the consumption of alcohol in C57BL/6J mice? Life Sci 40:2191–2194
Herrnstein RJ (1970) On the law of effect. J Exp Anal Behav 13:243–266
Holtzman SG, Villarreal J (1973) Operant behavior in the morphine-dependent rhesus monkey. J Pharmacol Exp Ther 184:528–541
Johanson CE (1984) Assessment of the dependence potential of cocaine in animals. In: Grabowski J (ed) Cocaine: pharmacology, effects and treatment of abuse. NIDA Res Monogr 50. US Government Printing Office, Washington, DC, pp 54–71
Johanson CE, Balster RL, Bonese K (1976) Self-administration of psychomotor stimulant drugs: the effects of unlimited access. Pharmacol Biochem Behav 4:45–51
Katz RJ, Baldrighi G, Roth K (1978) Appetitive determinants of self-stimulation. Behav Biol 23:500–508
Lester D, Greenberg LA (1952) Nutrition and the etiology of alcoholism: the effect of sucrose, saccharin and fat on the selfselection of ethyl alcohol by rats. O J Stud Alcohol 13:553–560
McMillan DE, Leander JD (1978) Chronic chlordiazepoxide and pentobarbital interactions on punished and unpunished behavior. J Pharmacol Exp Ther 207:515–520
Papasava M, Oei TPS, Singer G (1981) Low dose cocaine selfadministration of naive rats: Effects of body weights and a fixed-time one minute food delivery schedule. Pharmacol Biochem Behav 15:485–488
Rachlin H (1978) Contrast and matching. Psychol Rev 80:217–234
Samson HH, Falk JL (1974) Altration of fluid preference in ethanol-dependent animals. J Pharmacol Exp Ther 190:365–376
Samson HH, Lindberg K (1984) Comparison of sucrose-sucrose to sucrose-ethanol: concurrent responding in the rat: reinforcement schedule and fluid concentration effects. Pharmacol Biochem Behav 20:973–977
Samson HH, Roehrs TA, Tolliver GA (1982) Ethanol reinforced responding in the rat. A concurrent analysis using sucrose as the alternative choice. Pharmacol Biochem Behav 17:333–339
Schuster CR (1969) Variables affecting the self-administration of drugs by rhesus monkeys. In: Vagtborg H (ed) Use of nonhuman primates in drug evaluation. University of Texas Press, Austin, Tex, p 283
Siegel RK (1984) Changing patterns of cocaine use: longitudinal observation, consequences and treatment. NIDA Res Monogr 50. US Government Printing Office, Washington, DC, pp 92–110
Simpson DM, Annau Z (1977) Behavioral withdrawal following several psychoactive drugs. Pharmacol Biochem Behav 7:59–64
Slifer BL, Balster RL, Woolverton WL (1984) Behavioral dependence produced by continuous phencyclidine infusion in rhesus monkeys. J Pharmacol Exp Ther 230:339–406
Smith JC, Foster DF (1980) Some determinants of intake of glucose and saccharin solutions. Physiol Behav 25:127–133
Smith JC, Williams DP, Jue SS (1976) Rapid oral mixing of glucose and saccharin by rats. Science 191:304–305
Steinfels GF, Young GA (1981) Effects of narcotic abstinence on schedule-controlled behavior in dependent rats. Pharmacol Biochem Behav 14:393–395
Thompson T, Schuster CR (1964) Morphine self-administration, food-reinforced, and avoidance behaviors in rhesus monkeys. Psychopharmacologia 5:87–94
Valenstein ES, Cox VC, Kakolewski JW (1967) Polydipsia elicited by the synergistic action of a saccharin and glucose solution. Science 157:552–554
Weeks JR (1972) Long term intravenous infusion. In: Myers RD (ed) Methods in psychobiology, vol 2. Academic Press, London, pp 155–168
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carroll, M.E., Lac, S.T. & Nygaard, S.L. A concurrently available nondrug reinforcer prevents the acquisition or decreases the maintenance of cocaine-reinforced behavior. Psychopharmacology 97, 23–29 (1989). https://doi.org/10.1007/BF00443407
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00443407