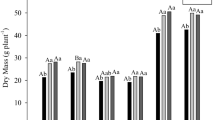
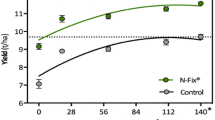
Abstract
A model system is described where Azospirillum and germinated wheat seeds were grown in association for a week and then assayed for nitrogen fixation (C2H2-reduction) and denitrification (N2O-formation) activities. The association performed C2H2-reduction and N2O-formation under microaerobic conditions. Both activities were measurable after already 3–5 h of incubation with substantial rates and were strictly dependent on the presence of both plants and bacteria. During the week of the growth of the association, the bacteria had lived exclusively from the carbon compounds supplied by the roots of the plants. C2H2-reduction activity by the association was more or less the same with all the Azospirillum brasilense strains, but lower with A. lipoferum and with the A. amazonense strains tested. Two nitrogenase negative mutants of Azospirillum brasilense showed virtually no activity in the association. C2H2-reduction activity was strongly dependent on the growth temperature of the association. Denitrification (N2O-formation) was high also at higher temperatures and at pH-values in the medium around 7.8 but not at neutrality and was strictly dependent on nitrate. The Azospirillum strain used strongly determined the rate of the N2O-formation in the association. It is suggested that Azospirillum may be beneficial to crops particularly under tropical conditions.
Similar content being viewed by others
References
Balderstone WL, Sherr B, Payne WJ (1976) Blockage by acetylene of nitrous oxide reduction in Pseudomonas perfectomarinus. Appl Environm Microbiol 31:504–508
Baldini JI, Döbereiner J (1984) Occurrence of Azospirillum amazonense in cereal roots. In: Abstract book of the third International Symposium on nitrogen fixation with non-legumes. Sitra Nitrogen Fixation Project, P.O. Box 329, SF-00121 Helsinki, Finland, p 64
Berg RH, Tyler ME, Novick NJ, Vasil V, Vasil JK (1980) Biology of Azospirillum sugarcane association: enhancement of nitrogenase activity. Appl Environm Microbiol 39:642–649
Bothe H, Klein B, Stephan MP, Döbereiner J (1981) Transformations of inorganic nitrogen by Azospirillum spp. Arch Microbiol 130:96–100
Bothe H, Barbosa G, Döbereiner J (1983a) Nitrogen fixation and nitrate respiration by Azospirillum brasilense. Z Naturforsch 38c:571–577
Bothe H, Yates MG, Cannon FC (1983b) Physiology, biochemistry and genetics of dinitrogen fixation. In: Läuchli A, Bieleski RL (eds) Encyclopedia of plant physiol, vol 15 A, Springer, Berlin Heidelberg New York Tokyo, pp 242–285
Cohen E, Okon Y, Kigel J, Nur I, Henis Y (1980) Increase in dry weight and total nitrogen content in Zea mays and Setaria italica associated with nitrogen-fixing Azospirillum spp. Plant Physiol 66:746–749
De-Polli H, Boyer CD, Neyra CA (1982) Nitrogenase activity associated with roots and stems of field grown corn (Zea mays L.) plants. Plant Physiol 70:1609–1613
Döbereiner J (1983) Dinitrogen fixation in rhizosphere and phyllosphere associations. In: Läuchli A, Bieleski R (eds) Encyclopedia of plant physiology, New Series, vol 15A. Springer, Berlin Heidelberg New York, pp. 330–350
Haahtela K, Wartiovaara T, Sundman V, Skujins J (1981) Root associated N2-fixation (C2H2-reduction) by enterobacteriaceae and Azospirillum strans in cold climate spodosols. Appl Environm Microbiol 41:203–206
Hartmann A, Singh A, Klingmüller W (1983) Isolation and characterization of Azospirillum mutants excreting high amounts of indoleacetic acid. Can J Microbiol 29:916–923
Heulin T, Bally R, Balandreau J (1982) Isolation of very efficient N2-fixing bacteria from the rhizosphere of rice. In: Klingmüller W (ed) Azospirillum genetics, physiology, ecology. Experientia, Suppl 42. Birkhäuser, Basel, pp 92–96
Jagnow G (1982) Growth and survival of Azospirillum lipoferum in soil and rhizosphere as influenced by ecological stress conditions. In: Klingmüller W (ed) Azospirillum genetics, physiology, ecology, Experientia, Suppl 42. Birkhäuser, Basel, pp 100–107
Jara P, Quiviger B, Laurent P, Elmerich C (1983) Isolation and genetic analysis of Azospirillum brasilense Nif- mutants. Can J Microbiol 29:968–972
Lin W, Okon Y, Hardy RWF (1983) Enhanced mineral uptake by Zea mays and Sorghum bicolor roots inoculated with Azospirillum brasilense. Appl Environm Microbiol 45:1775–1779
Martin P, Glatzle A (1982) Mutual influences of Azospirillum spp. and grass seedlings. In: Klingmüller W (ed) Azospirillum genetics, physiology, ecology. Experientia, Suppl 42. Birkhäuser, Basel, pp 108–120
Nelson LM, Knowles R (1978) Effect of oxygen and nitrate on nitrogen fixation and denitrification by Azospirillum brasilense grown in continuous culture. Can J Microbiol 24:1395–1403
Nur J, Okon Y, Henis Y (1982) Effect of dissolved oxygen tension on production of carotenoids, poly-β-hydroxybutyrate, succinate oxidase and superoxide dismutase by Azospirillum brasilense Cd grown in continuous culture. J Gen Microbiol 128:2937–2943
Okon Y, Heytler PG, Hardy RWF (1983) N2-fixation by Azospirillum brasilense and its incorporation into host Setaria italica. Appl Environm Microbiol 46:694–697
Pedrosa FO, Yates MG (1984) Regulation of nitrogen fixation (nif) genes of Azospirillum brasilense by nif A and ntr (gln) type gene products. FEMS Microbiol Letters 23:95–101
Penteado Stephan M, Zimmer W, Bothe H (1984) Denitrification by Azospirillum brasilense Sp 7. II. Growth with nitrous oxide as respiratory electron acceptor. Arch Microbiol 138:212–216
Rennie RJ, de Freitas JR, Morrison RJ (1984) Tracing dinitrogen (N2) fixation throughout the development of Canadian wheat. In: Abstract book of the third International Symposium on nitrogen fixation with non-legumes. Sitra Nitrogen Fixation Project, P.O. Box 329, SF-00121 Helsinki, Finland, p 36
Reynders L, Vlassak K (1982) Use of Azospirillum brasilense as biofertilizer in intensive wheat cropping. Plant and Soil 66:217–273
Schank SC, Weier KL, MacRae IC (1981) Plant yield and nitrogen content of a digitgrass in response to Azospirillum inoculation. Appl Environm Microbiol 41:342–345
Smith RL, Schank SC, Milam JR, Baltensperger AA (1984) Response of Sorghum and Penisetum species to the N2-fixing bacterium Azospirillum brasilense. Appl Environm Microbiol 47:1331–1336
Tibelius KH, Knowles R (1984) Uptake hydrogenase activity in denitrifying Azospirillum brasilense grown anaerobically with nitrous oxide or nitrate. J Bacteriol 157:84–88
Tien TM, Gaskins MH, Hubbell DH (1979) Plant growth substances produced by Azospirillum brasilense and their effect on the growth of pearl millet (Penisetum americanum L.). Appl Environm Microbiol 37:1016–1024
Van Berkum P, Bohlool BB (1980) Evaluation of nitrogen fixation by bacteria in association with roots of tropical grasses. Microbiol Rev 44:491–517
Volpon AGT, De-Polli H, Döbereiner J (1981) Physiology of nitrogen fixation in Azospirillum lipoferum Br 17 (ATCC 29709). Arch Microbiol 128:371–375
Yoshinari T, Knowles R (1976) Acetylene inhibition of nitrous oxide reduction by denitrifying bacteria Biochem Biophys Res Commun 69:705–710
Zimmer W, Penteado Stephan M, Bothe H (1984) Denitrification by Azospirillum brasilense Sp 7, I. Growth with nitrite as respiratory electron acceptor. Arch Microbiol 138:206–211
Zuberer DA, Alexander DB (1984) Effects of oxygen partial pressure and combined nitrogen on N2-fixation (C2H2-reduction) associated with Zea mays and other gramineous species. In: Abstract book of the third International Symposium on nitrogen fixation with non-legumes. Sitra Nitrogen Fixation Project, P.O. Box 329, SF-00121 Helsinki, Finland, p 20
Author information
Authors and Affiliations
Additional information
Dedicated to Professor Dr. Gerhart Drews, Freiburg, on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Neuer, G., Kronenberg, A. & Bothe, H. Denitrification and nitrogen fixation by Azospirillum . Arch. Microbiol. 141, 364–370 (1985). https://doi.org/10.1007/BF00428851
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00428851