Abstract
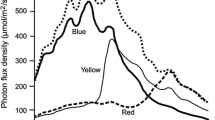
Trochophore larvae of Spirobranchus giganteus (Pallas) respond positively to white light at levels of illumination from 1 to 2 168×1014 quanta cm-2 s-1. In this range the strength of the response is not correlated with irradiance level. The response is increased by dark adaptation. At low levels of irradiance (0.1-2.0×1014 quanta cm-2 s-1) larvae respond positively to blue (360-510 nm, max. 430 nm) and green (475–620 nm, max. 530 nm) light but not to wavelengths of 590 nm or over. The light response develops gradually during the 12 h following the appearance of the eyespot and is maintained throughout the remainder of the planktonic phase.
Similar content being viewed by others
Literature cited
Bradley, D. J. and R. B. Forward Jr.: Phototaxis of adult brine shrimp, Artemia salina. Can. J. Zool. 62, 2357–2359 (1984)
Emery, A.R.: Eddy formation from an oceanic island: ecological effects. Carib. J. Sci. 12, 121–128 (1972)
Forward, R. B. Jr.: Light and diurnal vertical migration: photobehaviour and photophysiology of plankton. In: Photochemical and photobiological reviews, Vol. 1, pp 157–209. Ed. K. C. Smith. New York: Plenum Press 1976
Jackson, G. A. and R. R. Strathmann: Larval mortality from offshore mixing as a link between precompetent and competent periods of development. Am. Nat. 118, 16–26 (1981)
Lacalli, T.: Structure and organization of the nervous system in the trochophore larva of Spirobranchus. Phil. Trans. R. Soc. Lond. B. 306, 79–135 (1984)
Marsden, J. R.: Swimming in response to light by larvae of the tropical serpulid Spirobranchus giganteus. Mar. Biol. 83, 13–16 (1984)
Marsden, J. R. and D. T. Anderson: Larval development and metamorphosis of the serpulid polychaete Galeolaria caespitosa Lamarck. Aust. J. mar. freshwat. Res. 32, 667–680 (1981)
Murray, S. P., H. H. Roberts, D. M. Conlon and G. M. Rudder: Nearshore current fields around coral islands: control on sediment accumulation and reef growth. Proc. Third int. Coral Reef Symp., Miami, Fl. 1977
Scheltema, R. S. and A. H. Scheltema: Development, settlement and metamorphosis of Spirobranchus giganteus corniculatus (Grebe 1862). Summary of Research, Lizard Island Research Station 1978
Segrove, F.: The development of the serpulid Pomatoceros triqueter. Q. J. microsc. Sci. 82, 467–540 (1941)
Wisely, B.: The development and settling of the serpulid worm Hydroides norvegica Gunnerus (Polychaeta). Aust. J. mar. freshwat. Res. 9, 351–361 (1958)
Young, C. M. and F.-S. Chia: Ontogeny of phototaxis during larval development of the sedentary polychaete, Serpula vermicularis (L.). Biol. Bull. mar. biol. Lab., Woods Hole 162, 457–468 (1982)
Author information
Authors and Affiliations
Additional information
Communicated by R. W. Doyle, Halifax
Rights and permissions
About this article
Cite this article
Marsden, J.R. Response to light by trochophore larvae of Spirobranchus giganteus . Marine Biology 93, 13–16 (1986). https://doi.org/10.1007/BF00428649
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00428649