Abstract
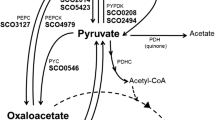
The mechanism of the massive extracellular production of citric and isocitric acids by Saccharomycopsis lipolytica grown on n-paraffins has been studied. When growth stops, because of nitrogen limitation, the intracellular concentration of ATP sharply rises whereas that of AMP and ADP decreases to a low level. At the same time production of acids begins. The activity of the NAD-dependent isocitrate dehydrogenase which requires AMP for activity becomes very low and prevents the oxidative function of the citric acid cycle whereas isocitrate lyase is not inhibited. As citrate synthase inhibition by ATP appears to be insufficient to stop n-paraffin degradation, citric and isocitric acids accumulation can take place. Massive excretion of these acids, however, probably still involves other physiological changes brought about by nitrogen limitation, possibly some permeabilization of the cell to these acids.
Similar content being viewed by others
References
Atkinson, D. E.: Enzymes as control elements in metabolic regulation. In: The enzymes, Vol. 1 (P. D. Boyer, ed.), pp. 461–489. New York-London: Academic Press 1970
Atkinson, D. E., Hathaway, J. A., Smith, E. C.: Kinetics of regulatory enzymes. Kinetic order of the yeast diphosphopyridine nucleotide isocitrate dehydrogenase reaction and a model for the reaction. J. biol. Chem. 240, 2682–2690 (1965)
Demain, A. L., Birnbaum, J.: Alteration of permeability for the release of metabolites from the microbial cell. Curr. Top. Microbiol. Immunol. 46, 1–25 (1968)
Dixon, G. H., Kornberg, H. L: Assay methods for key enzymes of the glyoxylate cycle. Biochem. J. 72, 3 p. (1959)
Hathaway, J. A., Atkinson, D. E.: The effect of adenylic acid on yeast nicotinamide adenine dinucleotide isocitrate dehydrogenase, a possible metabolic control mechanism. J. biol. Chem. 238, 2875–2881 (1963)
Hathaway, J. A., Atkinson, D. E.: Kinetics of regulatory enzymes: Effect of adenosine triphosphate on yeast citrate synthase. Biochem. biophys. Res. Commun. 20, 661–665 (1965)
Hildebrandt, W., Weide, H.: Isocitratlyase von Candida guilliermondii, Stamm 17. I. Reinigung und Charakterisierung. Z. allg. Mikrobiol. 13, 569–576 (1973)
Hildebrandt, W., Weide, H.: Isocitratlyase von Candida guilliermondii, Stamm H 17. II. Regelung durch ausgewählte Intermediate des Tricarbonsäurezyclus und Hexoseabbaus. Z. allg. Mikrobiol. 14, 39–46 (1974)
Javorek, D., Gruber, W., Bergmeyer, H. U.: Adenosine-5′-triphosphate. In: Methods of enzymatic analysis, Vol. 4 (H. U. Bergmeyer, ed.), pp. 2097–2101. Weinheim: Verlag Chemie; New York-London: Academic Press 1974a
Javorek, D., Gruber, W., Bergmeyer, H. U.: Adenosine-5′-diphosphate and adenosine-5′-monophosphate. In: Methods of enzymatic analysis, Vol. 4 (H. U. Bergmeyer, ed.), pp. 2127–2131. Weinheim: Verlag Chemie; New York-London: Academic Press 1974b
Layne, E.: Spectrophotometric and turbidimetric methods for measuring proteins. In: Methods in enzymology, Vol. 3 (S. P. Colowick, N. O. Kaplan, eds.), pp. 447–454. New York: Academic Press 1957
Marchal, R.: Contribution à l'étude de l'excrétion d'acide citrique par une levure du genre Candida se développant sur n-alcanes. Thèse de spécialité, University of Nancy (1975)
Parvin, R., Atkinson, D. E.: Purification and some properties of yeast citrate synthase. Arch. Biochem. Biophys. 128, 528–533 (1968)
Ramaley, R. F., Hudock, M. O.: Purification and properties of isocitrate dehydrogenase (NADP) from Thermus aquaticus YT-1, Bacillus subtilis 168 and Chlamydomonas reinhardii Y-2. Biochim. biophys. Acta (Amst.) 315, 22–36 (1973)
Weitzman, P. D. J., Hewson, J. K.: In situ regulation of citrate synthase. Absence of ATP inhibition observed in vitro. FEBS Letters 36, 227–231 (1973)
Author information
Authors and Affiliations
Additional information
This work is a part of a “Doctorat de Spécialité” Thesis submitted by R. Marchal to the University of Nancy (1975)
Rights and permissions
About this article
Cite this article
Marchal, R., Vandecasteele, JP. & Metche, M. Regulation of the central metabolism in relation to citric acid production in Saccharomycopsis lipolytica . Arch. Microbiol. 113, 99–104 (1977). https://doi.org/10.1007/BF00428588
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00428588