Abstract
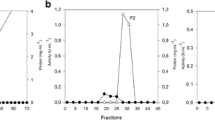
It was found that crude preparation obtained from the culture medium of Fusarium avenaceum degraded cellulose and xylan. After chromatography on CM-Sepharose CL-6B of this preparation six fraction were obtained. The eluted fractions II and V showed xylanase activity, fraction IV — cellulase activity and fraction III — xylanase and cellulase activity. The end products of xylan hydrolysis by all xylanase fractions (II, III, V) were xylobiose, xylose, xylotriose and xylotetrose. The end products of cellulose hydrolysis by fractions III and IV was cellobiose, glucose and cellotriose. The data from gel filtration on Sephacryl S-200 indicated a molecular weight of more than 250,000 for both cellulase IV and xylanase V. After gel filtration in the presence of urea disaggregation of those high molecular xylanase and cellulase particles was observed. Xylanase II in difference from the other fractions contained higher amount of sugar. Digestion of fraction II with cellulase-hemicellulase preparation from Phoma hibernica decreased the content of sugar from 17% to 8%, but did not change its enzymatic properties. Cellulase IV as well as xylanase V were inactivated by N-bromosuccinimide, 2-hydroxy-5-nitrobenzyl bromide and tetranitromethane, hence it is suggested that tryptophan and tyrosine are the essential for the activity of these enzymes.
Similar content being viewed by others
References
Cooper RM, Wood RKS (1975) Regulation of synthesis of cell wall degrading enzymes by Verticillium alboatrum and Fusarium oxysporum f. sp. lycopersici. Physiol Plant Pathol 5:135–156
Davis BJ (1964) Disc electrophoresis. II: Method and application to human serum proteins. Ann NY Acad Sci 121:404–427
Dever JE, Jr, Bandurski RS, Kivilaan A (1968) Partial chemical characterization of corn root cell walls. Plant Physiol 43:50–56
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determinations of sugars and related substances. Anal Chem 28:350–356
Gum EK Jr, Brown RD Jr (1977) Comparison of four purified extracellular 1,4-β-d-glucan cellobiohydrolase enzymes from Trichoderma viride. Biochim Biophys Acta 492:225–231
Hurst PL, Sullivan PA, Shephard MG (1977) Chemical modification of a cellulase from Aspergillus niger. Biochem J 167:549–556
Kanda T, Wakabayashi K, Nisizawa K (1976) Purification and properties of an endo-cellulase of Avicelase type from Irpex lacteus. J Biochem 79:977–988
Montant Ch, Thomas L (1977) Structure d'un glucane exocellulaire produit par le Botrytis cinerea (Pers). Ann Sci Naturelles Bot 18:185–192
Mullen JM, Bateman DF (1975) Polysaccharide degrading enzymes produced by Fusarium roseum “Avenaceum” in culture and during pathogenesis. Physiol Plant Pathol 6:233–246
Nakayama M, Tomita Y, Suzuki H, Nisizawa K (1976) Partial proteolysis of some cellulase components from Trichoderma viride and the substrate specificity of the modified products. J Biochem 79:955–966
Nelson N (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Urbanek H, Zalewska-Sobczak J, Borowinska A (1978) Isolation and properties of extracellular cellulase-hemicellulase complex of Phoma hibernica. Arch Microbiol 118:265–269
Walseth CS (1952) Occurence of cellulases in enzyme preparations from microorganisms. Tappi 35:228–232
Wood TM (1971) The cellulase of Fusarium solani; Purification and specificity of the β-(1 → 4)-glucanase and the β-d-glucosidase components. Biochem J 121:353–362
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zalewska-Sobczak, J., Urbanek, H. Cellulose and xylan degrading enzymes of Fusarium avenaceum . Arch. Microbiol. 129, 247–250 (1981). https://doi.org/10.1007/BF00425259
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00425259