Abstract
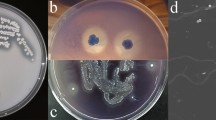
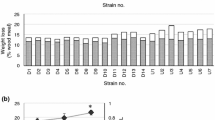
The lignin-degrading actinomycete Streptomyces viridosporus T7A readily degrades the lignin model compound dehydrodivanillin. Four mutants of this organism (produced by irradiation of spores with ultraviolet light) were shown to have lost the ability to catabolize dehydrodivanillin. These mutant strains retained an undiminished ability to degrade Douglas-fir lignin (14C-lignin → 14CO2) as compared to the wild-type strain. None of the strains accumulated detectable quantities of dehydrodivanillin when grown on lignocellulose. Thus it appears that the enzymes involved in dehydrodivanillin catabolism are not a part of the streptomycete's system for degrading polymeric lignin. It is concluded that dehydrodivanillin is probably not a relevant model compound for study of lignin polymer degradation by Streptomyces viridosporus. Since many stable mutants completely lacking DHDV-degrading ability were readily obtained, it is suggested that the relevant catabolic enzymes may be encoded on a plasmid.
Similar content being viewed by others
Abbreviations
- DHDV:
-
dehydrodivanillin
References
Adler E (1977) Lignin chemistry — past, present and future. Wood Sci Technol 11:169–218
Ander P, Eriksson K-E (1977) The importance of phenol oxidase activity in lignin degradation by the white-rot fungus Sporotrichum pulverulentum. Arch Microbiol 109:1–8
Cain RB (1980) The uptake and catabolism of lignin-related aromatic compounds and their regulation in microorganisms. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin biodegradation: Microbiology, chemistry, and potential applications, vol I. CRC Press, Inc, Boca Raton, Fl, pp 21–66
Chang H-m, Chen C-L, Kirk TK (1980) Chemistry of lignin degraded by white-rot fungi. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin biodegradation: Microbiology, chemistry, and potential applications, vol I. CRC Press, Inc, Boca Raton, Fl, pp 215–230
Crawford DL (1978) Lignocellulose decomposition by selected Streptomyces strains. Appl Environm Microbiol 35:1041–1045
Crawford DL, Crawford RL (1976) Microbial degradation of lignocellulose: the lignin component. Appl Environm Microbiol 31:714–717
Crawford RL, Crawford DL (1978) Radioisotopic methods for the study of lignin biodegradation. Dev Ind Microbiol 9:35–49
Crawford DL, Crawford RL (1980) Microbial degradation of lignin. Enzyme Microb Technol 2:11–22
Crawford RL, McCoy E, Harkin JM, Kirk TK, Obst JR (1973) Degradation of methoxylated benzoic acids by a Nocardia from a lignin-rich environment: significance to lignin degradation and effect of chloro substituents. Appl Microbiol 26:176–184
Crawford RL, Robinson L., Chen A (1980) C-Labeled lignins as substrates for the study of lignin biodegradation and transformation. In: Kirk TG, Higuchi T, Chang H-m (eds) Lignin Biodegradation: Microbiology chemistry, and potential applications, vol. I. CRC Press, Inc., Boca Raton, Fl pp 61–76
Elbs K, Lerch H (1976) Über Dehydrodivanillin. J. Prakt Chem 201:1–9
Fukuzumi T (1980) Microbial metabolism of lignin-related aromatics. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin Biodegradation: Microbiology, chemistry, and potential applications, vol. I. CRC Press, Inc., Boca Raton, Fl pp 61–76
Elbs K, Lerch H (1976) Über Dehydrodivanillin. J. Prakt Chem 201:1–9
Fukuzumi T (1980) Microbial metabolism of lignin-related aromatics. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin biodegradation: Microbiology, chemistry, and potential applications, vol. I. CRC Press, Boca Raton, Fl pp 73–94
Gold MH, Cheng TM (1978) Induction of colonial growth and replica plating of the white-rot basidiomycete Phanerochaete chrysosporium. Appl Environm Microbiol 35:1223–1225
Gold M, Cheng T, Krisnangkura K, Mayfield M, Smith L (1980) Genetic and biochemical studies on P. chrysosporium and their relation to lignin degradation. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin biodegradation: Microbiology chemistry, and potential applications, vol. II. CRC Press, Inc., Bota Raton, Fl pp 65–71
Gordon RE, Haynes WC, Pang CH-n (1973) The genus Bacillus. Agriculture handbook no. 427. GS Government Printing Office, Washington, DC
Harkin JM (1967) Lignin — a natural polymeric product of phenol oxidation. In: Battersby AR, Taylor WI (eds) Oxidative coupling of phenols. Marcel Dekker, Inc., New York, pp 243–321
Higuchi T (1980) Microbial degradation of dilignols as lignin models. In: Kirk TK, Higuchi T, Chang H-m (eds) Lignin biodegradation: Microbiology, Chemistry, and potential applications, vol. I. CRC Press, Inc., Boca Raton, Fl pp 171–193
Higuchi T, Shimada M, Nakatsubo F, Tanahashi M (1977) Differences in biosynthesis of guaiacyl and syringul lignins in woods. Wood Sci Technol 11:153–167
Kirk TK, Chang H-m (1974) Decomposition of lignin by white-rot fungi. I. Isolation of heavily degraded lignins from decayed spruce. Holzforschung 28:217–222
Kirk TK, Chang H-m (1975) Decomposition of lignin by white-rot fungi. II. Characterization of heavily degraded lignins from decayed spruce. Holzforschung 29:56–64
Lundquist K, Kirk TK (1971) Acid degradation of lignin. IV. Analysis of lignin acidolysis products by gas chromatography, using trimethylsilyl derivatives. Acta Chem Scand 25:889–894
Moore WE, Johnson DB (1967) Procedures for the analysis of wood and wood products. U.S.D.A. Forest Service, Forect Products Laboratory, Madison, WI
Phelan MB, Crawford DL, Pometto AL, III, (1979) Isolation of lignocellulose-decomposing actinomycetes and degradation of specifically 14C-labeled lignocellulose by six selected Streptomyces strains. Can J Microbiol 25:1270–1276
Redshaw PA, McCann PA, Sankaran L, Pogell BM (1976) Control of differentiation in Streptomycetales: involvement of extrachromosomal deoxyribonucleic acid and glucose repression in aerial mycelia development. J Bacteriol 125:698–705
Sarkanen KV, Ludwig CH (eds), Lignins: Occurrence, formation, structure, and reactions. Wiley-Interscience NY
Suter M, Hütter R, Leisinger Th (1978) Mutants of Streptomyces glaucescens affected in the production of extracellular enzymes. In: Freerksen E, Tarnok I, Thumin JH (eds) Genetic of the actinomycetales —Proceedings of the International Colloquium at the Forschungsinstitut Borstel. Gustav Fischer Verlag, Stuttgart
Vivan A (1971) Genetic control of fertility in Streptomyces coelicolor A3(2): plasmid involvement in the interconversion of UF and IF strains. J Gen Microbiol 69:353–364
Weinstein DA, Krisnangkura K, Mayfield MB, Gold MG (1980) Metabolism of radiolabeled β-guaiacyl ether linked lignin dimeric compounds by Phanerochaete chrysosporium. Appl Environm Microbiol 39:535–540
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crawford, R.L., Crawford, D.L. & Dizikes, G.J. Catabolism of the lignin substructure model compound dehydrodivanillin by a lignin-degrading Streptomyces . Arch. Microbiol. 129, 204–209 (1981). https://doi.org/10.1007/BF00425251
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00425251