Summary
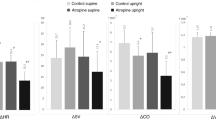
Endogenous opiate peptides are known to exert a depressant action on ventilation (\(\dot V_E \)), and their plasma levels have been shown to be elevated during a variety of exercise protocols. We investigated whether they might modulate the control of the hyperpnea of short-term constant-load (CLE) and incremental (IE) cycle-ergometer exercise. Four healthy subjects performed CLE tests at ca. 80% of the anaerobic threshold (θan) for 5 min following a period of unloaded pedaling, and IE tests (10 or 20 W min−1) to the limit of tolerance. Normal saline (3 ml) or the opiate antagonist naloxone (1.2 mg in 3 ml) were administered intravenously prior to each test. Naloxone elicited no discernible effect on \(\dot V_E \), alveolar gas tensions, or heart rate throughout the entire range of work rates; neither were θan nor the maximum work rate affected. It is concluded that, for short-term exercise ranging in intensity from moderate to severe, the role played by endogenous opiate peptides in the control of the exercise hyperpnea appears to be neglible in man.
Similar content being viewed by others
References
Atweh SF, Kuhar MJ (1977) Autoradiographic localization of opiate receptors in rat brain. (1) Spinal cord and lower medulla. Brain Res 124:53–67
Atweh SF, Murrin LC, Kuhar MJ (1978) Presynaptic localization of opiate receptors in the vagal and accessory optic systems; an autoradiographic study. Neuropharmacol 17:65–71
Brooks S, Burrin J, Cheetham ME, Hall G, Williams C, Yeo T (in press) The Β-endorphin response to brief maximal exercise in man. J Physiol (Lond)
Carr DB, Bullen BA, Skriner GS, Arnold MA, Rosenblatt M, Beitins IZ, Martins JB, McArthur JW (1981) Physical conditioning facilitates the exercise-induced secretion of beta-endorphin and 2beta- lipotropin in women. N Eng J Med 305:560–563
Catlin DH, Hui KK, Loh HH, Li CH (1977) Pharmacologic activity of beta endorphin in man. Comm Psychopharmacol 1:439–500
DuBois A, Britt AG, Fenn WO (1951) CO2 during the respiratory cycle. J Appl Physiol 4:535–548
Fleetham JA, Clark H, Dhingra S, Chernick V, Anthonisen NR (1980) Engogenous opiates and the chemical control of breathing in humans. Am Rev Respirat Dis 121:1045–1051
Florez JA, Mediavilla A, Pazos A (1980) Respiratory effects of Β-endorphin, D-ALA2-met-enkephalinamide, and met-enkephalin injected into the lateral ventricle and the ponto-medullary subarachnoid space. Brain Res 199:197–206
Fraioli F, Moretti C, Paolucci D, Alicicco E, Crescenzi F, Fortunio G (1980) Physical exercise stimulates marked concomitant release of beta-endorphin and adrenocorticotropic hormone (ACTH) in peripheral blood in man. Experientia 36:987–989
Grimby G, Saltin B, Wilhelmsen L (1971) Pulmonary flowvolume and pressure-volume relationship during submaximal and maximal exercise in young well-trained men. Bull Eur Physiopathol Respir 7:157–167
Grossman A, Clement-Jones V (1983) Opiate receptors: enkephalins and endorphins. Clin Endocrinol Metab 12:31–56
Grossman A, Bouloux P, Price P, Drury PL, Lam KSL, Turner T, Thomas J, Besser GM, Sutton J (1984) The role of opioid peptides in the hormonal responses to acute exercise in man. Clin Sci 67:483–491
Hokfeldt T, Elde R, Johansson O, Terenius L, Stein L (1977) The distribution of enkephalin immunoreactive cell bodies in the rat central nervous system. Neurosci Lett 5:25–31
Jaffe JH, Martin WR (1975) Narcotic analgesics and antagonists. In: Goodman LH, Gilman A (eds) The pharmacological basis of therapeutics. MacMillan, New York, pp 274–276
Kaufman RD, Gabathuler ML, Bellville JW (1981) Potency, duration of action and pA2 in man of intravenous naloxone measured by reversal of morphine-depressed respiration. J Pharmacol Exp Ther 219:156–162
Lawson EE, Waldrop TG, Eldridge FL (1979) Naloxone enhances respiratory output in cats. J Appl Physiol 47:1105–1111
McMurray RG, Sheps DS, Guinan DM (1984) Effects of naloxone on maximimal stress testing in females. J Appl Physiol 56:436–440
McQueen DS, Ribeiro JA (1981) Effects of Β-endorphin, vasoactive intestinal polypeptide and cholecystokinin octapeptide on cat carotid chemoreceptor activity. Quart J Exp Physiol 66:273–284
Martin BJ, Zwillich CW, Weil JV (1980) Morphine reduces ventilation without changing metabolic rate in exercise. Med Sci Sports Exer 12:285–287
Moss IR, Scarpelli EM (1981) Beta-endorphin central depression of respiration and circulation. J Appl Physiol 50:1011–1016
Ngai SH, Berkowitz BA, Yang JC, Hempstead J, Spector S (1976) Pharmacokinetics of naloxone in rats and in man: basis for its potency and short duration of action. Anesthesiology 44:398–401
Pokorski M, Lahiri S (1981) Effects of naloxone on carotid body chemoreception and ventilation in the cat. J Appl Physiol 51:1533–1538
Pokorski M, Grieb P, Wideman J (1981) Opiate system influences central respiratory chemosensors. Brain Res 211:221–226
Rossier J, French ED, Rivier C, Ling N, Guillemin R, Bloom FE (1977) Footshock-induced stress increases beta-endorphin levels in blood but not brain. Nature (Lond) 270:618–620
Santiago TV, Johnson J, Riley DJ, Edelman NH (1979) Effects of morphine on ventilatory response to exercise. J Appl Physiol 47:112–118
Santiago TV, Remolina C, Scoles V, Edelman NH (1981) Endorphins and the control of breathing. N Engl J Med 304:1109–1195
Sessle BJ, Henry JL (1985) Effects of enkephalin and 5-hydroxytryptamine on solitary tract neurones involved in respiration and respiratory reflexes. Brain Res 327:221–230
Walmsley JK, Zarbin MA, Young WS, Kuhar MJ (1982) Distribution of opiate receptors in the monkey brain: an auto-radiographic study. Neuroscience 7:595–613
Wasserman K, Whipp BJ (1975) Exercise physiology in health and disease. Am Rev Respirat Dis 112:219–249
Wasserman K, Whipp BJ, Casaburi R, Beaver WL, Brown HV (1977) CO2 flow to the lungs and ventilatory control. In: Dempsey JA, Reed CE (eds) Muscular exercise and the lung. Univ Wisconsin Press, Madison, pp 103–135
Whipp BJ (1981) The control of exercise hyperpnea. In: Hornbein T (ed) The regulation of breathing. Dekker, New York, pp 1069–1139
Whipp BJ, Ward SA, Davis JA, Wasserman K (1984) Estimating mean arterial PCO2 from alveolar CO2 profiles during exercise in man. Fed Proc 43:634
Willer JC, Boureau F, Dauthier C, Bonora M (1979) Study of naloxone in normal awake man: effects on heart rate and respiration. Neuropharmacology 18:469–472
Wilson BA, Eldridge FL, Millhorn DE, Waldrop TG, Klingler LE (1984) Effects of centrally administered naloxone on phrenic nerve activity. Brain Res 324:366–368
Author information
Authors and Affiliations
Additional information
Senior Investigator of the American Heart Association (Greater Los Angeles Affiliate)
Rights and permissions
About this article
Cite this article
Griffis, C., Kaufman, R.D. & Ward, S.A. Naloxone and the ventilatory response to exercise in mana. Europ. J. Appl. Physiol. 55, 624–629 (1986). https://doi.org/10.1007/BF00423207
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00423207