Summary
The tissue distribution of different glutathione S-transferases (GST) is analysed by electrophoresis.
-
1)
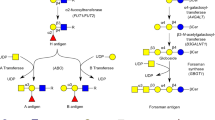
The existence of GST“e” (erythrocyte), GST3, GST1, and GST2 is confirmed. GST“e” the fastest and most thermolabile of different GST analysed is observed only in erythrocyte cells. GST3 which migrates more slowly than GST“e” is present in all tissues and cells analysed, excepted for erythrocyte cells in which only GST“e” is observed. GST1 presents a polymorphism with four phenotypes 1, 1/2, 2, and 0 controlled by three alleles 1, 2, and 0 (null). With the sample of 56 livers analysed the different frequencies obtained are 9%, 5%, 43%, 43% for the phenotypes 1, 1/2, 2, and 0 respectively and 0.074 (p), 0.279 (q), 0.647 (r) for the alleles 1, 2, and 0 (null). GST2 presents variant patterns due probably, in the majority of cases, to post-synthetic modifications rather than allelic variation.
-
2)
Two new GST are described, GST4 and GST5. GST4 abundant in muscle tissue is a dimeric protein. GST4 forms with GST1 a heterodimeric band. GST5 is observed in brain homogenates.
-
3)
For the chromosome localization the results obtained by man (leucocytes)-mouse somatic cell hybrid analysis indicate that the gene for leucocytes GST is on chromosome 11. This gene is the structural GST3 gene.
Similar content being viewed by others
References
Awasthi YC, Dao DD, Partridge CA (1981) Genetic origin of human glutathione S-transferases. Am J Hum Genet 33:35A
Board PG (1981) Biochemical genetics of glutathione S-transferase in man. Am J Hum Genet 33:36–43
Boyland E, Chasseaud LF (1969) The role of glutathione and glutathione S-transferase in mercapturic acid biosynthesis. Adv Enzymol 32:173–219
Buttin G, Le Guern G, Phalente L, Lin ECC, Medrano L, Cazenave PA (1978) Lymphocyte hybridomas. Curr Topics Microbiol Immunol 81:27–36
Chasseaud LF (1976) In: Arias IM, Jakoby WB (eds) Glutathione: metabolism and function. Raven Press, New York, pp 243–257
Dao DD, Partridge CA, Awasthi YC, Srivastava SK (1982) Interrelationship between glutathione S-transferases of human liver and placenta. Am J Hum Genet 34:49A
Davidson RG, Corner JA, Rattazzi MC (1970) Genetic polymorphism of human mitochondrial glutamic oxaloacetic transaminase. Science 169:391–392
Dutrillaux B, Lejeune J (1971) Sur une nouvelle technique d'analyse du caryotype humain. C R Acad Sci (D) (Paris) 272:2638–2640
Guthenberg C, Akerfeldt K, Mannervik B (1979) Purification of glutathione S-transferase from human placenta. Acta Chem Scand [8] 33:595–596
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Kamisaka K, Habig WH, Ketley JN, Arias IM, Jakoby WB (1975) Multiple forms of human glutathione S-transferase and their affinity for bilirubin. Eur J Biochem 60:153–161
Ketley JN, Habig WH, Jakoby WB (1975) Binding of nonsubstrate ligands to the glutathione S-transferase. J Biol Chem 250:8670–8673
Koskelo K, Valmet E (1980) Acid glutathione S-transferase from human liver: preliminary report. Scand J Clin Lab Invest 40:179–184
Laisney V, Nguyen Van Cong, Gross MS, Parisi I, Foubert C, Weil D, Frezal J (1983) Localisation du groupe synténique LDHA-GST3-ESA4 sur le chromosome 11 chez l'homme. Analyses des hybrides homme-rongeur classiques et d'un type nouveau (non adhérents à la paroi). Ann Génét (Paris) 26:69–74
Litwack G, Ketterer B, Arias IM (1971) Ligandin: a hepatic protein which binds steroïds, bilirubin, carcinogens and a number of exogenous organic anions. Nature 234:466–467
Marcus CJ, Habig WH, Jakoby WB (1978) Glutathione transferase from human erythrocytes. Nonidentity with the enzymes from liver. Arch Biochem Biophys 188:287–293
Meera Khan P (1971) Enzyme electrophoresis on cellulose acetate gel: zymogram pattern in man-mouse and man-Chinese hamster somatic cell hybrids. Arch Biochem Biophys 145:470–483
Nguyen Van Cong, Laisney V, Gross MS, Frezal J (1984) Glutathione S-transferases. Tissues distribution. Number of loci. Polymorphism. Chromosome localization. Los Angeles Conference (1983). Human Gene Mapping 7. Birth Defects 20:554
Partridge CA, Dao DD, Awasthi YC (1982) Purification and properties of human lung GSH S-transferases. Am J Hum Genet 34:59A
Pontecorvo G (1975) Production of mammalian somatic cell hybrids by mean of polyethylene glycol treatment. Somatic Cell Genet 1:397–400
Scott EM, Wright RC (1980) Variability of glutathione S-transferase of human erythrocytes. Am J Hum Genet 32:115–117
Shulman M, Wide CD, Kohler G (1978) A better cell line for making hybridomas secreting specific antibodies. Nature 276:269–270
Silberstein DL, Sakaguchi AY, Shows TB (1982) Assignment of the gene for glutathione S-transferase 1 (GST1) to human chromosome 11 Oslo Conference (1981) Human Gene Mapping 6. Birth Defects 18:317
Silberstein DL, Shows TB (1981) The gene for glutathione S-transferase 1 (GST1) is on human chromosome 11. Somatic Cell Genet 8:667–675
Strange RC, Faulder CG, Davis BA, Hume R, Brown JAH, Cotton W, Hopkinson DA (1984) The human glutathione S-transferases: studies on the tissue distribution and genetic variation of the GST1, GST2 and GST3 isozymes. Ann Hum Genet 48:11–20
Wood JL (1970) In: Fishman WH (ed) Metabolic conjugation and metabolic hydrolysis, vol 2. Academic Press, New York, pp 261–299
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Laisney, V., Van Cong, N., Gross, M.S. et al. Human genes for glutathione S-transferases. Hum Genet 68, 221–227 (1984). https://doi.org/10.1007/BF00418392
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00418392