Summary
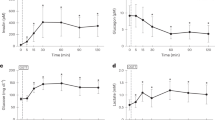
To assess the relative contributions of gluconeogenesis and glycogenolysis to overall hepatic glucose output in postabsorptive normal humans and those of the indirect and direct pathways for glycogen synthesis, we studied six normal volunteers, who had been fasted for 16 h to reduce their hepatic glycogen stores, and then ingested glucose (250 g over 10 h) enriched with [6-3H] glucose to replenish and label their hepatic glycogen. After a 10-h overnight fast, release of the [6-3H] glucose into the circulation was traced with [2-3H] glucose to estimate breakdown of glycogen that had been formed via the direct pathway while gluconeogenesis was simultaneously estimated by incorporation of infused [14C] lactate into plasma glucose. We found that release of [6-3H] glucose into plasma (6.79±0.69 μmol · kg−1 · min−1) accounted for 46±5% of hepatic glucose output (15.0±0.7 μmol · kg−1· min−1) while glucose formed from lactate (2.71±0.28 μmol · kg−1 · min−1) accounted for 19±2% of hepatic glucose output. Since these determinations underestimate direct pathway glycogenolysis and overall gluconeogenesis, a maximal estimate for the contribution of indirect pathway glycogenolysis to hepatic glucose output is obtained by subtracting the sum of direct pathway glycogenolysis and lactate gluconeogenesis from hepatic glucose output. This amounted to a maximum of 36±5 % of hepatic glucose output and 44±6% of overall glycogenolysis. Assuming that the relative proportions of direct and indirect pathway glycogen breakdown would reflect the relative contributions of these pathways to glycogen formation, we conclude that under our experimental conditions the direct pathway is the predominant route for glycogen formation in man and that in overnight fasted humans, hepatic glucose output is mainly the result of glycogenolysis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dietze G, Wicklmayr M, Hepp K, et al (1976) On gluconeogenesis of human liver: accelerated hepatic glucose formation by increased precursor supply. Diabetologia 12: 555–561
Wahren J, Felig P, Cerasi E, Luft R (1972) Splanchnic and peripheral glucose and amino acid metabolism in diabetes mellitus. J Clin Invest 51: 1870–1878
Consoli A, Kennedy F, Miles J, Gerich J (1987) Determination of Krebs cycle metabolic carbon exchange in vivo and its use to estimate gluconeogenesis in man. J Clin Invest 80: 1303–1310
Consoli A, Nurjhan N, Capani F, Gerich J (1989) Predominant role of gluconeogenesis in increased hepatic glucose production in NIDDM. Diabetes 38: 550–561
Nilsson L, Hultman E (1973) Liver glycogen in man — the effect of total starvation or a carbohydrate-poor diet followed by refeeding. Scand J Lab Invest 32: 325–330
Rothman D, Magnusson I, Katz L, Shulman R, Shulman G (1991) Quantitation of hepatic glycogenolysis and gluconeogenesis in fasting humans with 13C NMR. Science 254: 573–575
Magnusson I, Rothman D, Katz L, Shulman R, Shulman G (1992) Increased rate of gluconeogenesis in type II diabetes. A 13C nuclear magnetic resonance study. J Clin Invest 90: 1323–1327
Shulman G, Landau B (1992) Pathways of glycogen repletion. Physiol Rev 72: 1019–1035
McGarry J, Kuwajima M, Newgard C, Forster D, Katz J (1987) From dietary glucose to hepatic glycogen: the full circle round. Ann Rev Nutr 7: 51–73
Mitrakou A, Jones R, Okuda Y, et al (1991) Pathway and carbon sources for hepatic glycogen repletion in the dog. Am J Physiol 260: E194-E202
Moore M, Cherrington A, Cline G, et al (1991) Source of carbons for hepatic glycogen synthesis in the conscious dog. J Clin Invest 88: 578–587
Magnusson I, Chandramouli V, Schumann W, Kumaran K, Wahren J, Landau B (1987) Quantitation of the pathways of hepatic glycogen formation on ingesting a glucose load. J Clin Invest 80: 1748–1754
Magnusson I, Chandramouli V, Schumann W, Kumaran K, Wahren J, Landau B (1989) Pathways of hepatic glycogen formation in humans following ingestion of a glucose load in the fed state. Metabolism 38: 583–585
Radziuk J (1982) Sources of carbon in hepatic glycogen synthesis during absorption of an oral glucose load in humans. Fed Proc 41: 111–117
Consoli A, Nurjhan N, Gerich J (1989) Rate of appearance and disappearance of plasma lactate after oral glucose: implications for indirect-direct pathway hepatic glycogen repletion in man. Clin Physiol Biochem 7: 70–78
Radziuk J (1989) Hepatic glycogen in humans. I. direct formation after oral and intravenous glucose or after a 24-h fast. Am J Physiol 257: E145-E157
Radziuk J (1989) Hepatic glycogen in humans. II. Gluconeogenetic formation after oral and intravenous glucose. Am J Physiol 257: E158-E169
Shulman G, Cline G, Schumann W, Chandramouli V, Kumaran K, Landau B (1990) Quantitative comparison of pathways of glycogen repletion in fed and fasted humans. Am J Physiol 259: E335–341
Wajgnot A, Chandramouli V, Schumann W, Kumarin K, Efendic S, Landau B (1989) Test of the assumptions made in estimating the extent of futile cycling. Am J Physiol 256: E668-E675
Kreisberg RA, Pennington LF, Boshell BR (1970) Lactate turnover and gluconeogenesis in normal and obese humans. Diabetes 19: 53–63
Bloom B (1962) The simultaneous determination of C14 and H3 in the terminal groups of glucose. Anal Biochem 3: 85–87
Chiasson JL, Liljenquist JE, Lacy WW, Jennings AS, Cherrington AD (1977) Gluconeogenesis: methodological approaches in vivo. Fed Proc 36: 229–235
Bell P, Firth R, Rizza R (1989) Assessment of the postprandial pattern of glucose metabolism in nondiabetic subjects and patients with noninsulin-dependent diabetes mellitus using a simultaneous infusion of [2–3H] and [3-3H]- glucose. Metabolism 38: 38–45
Kelley D, Mitrakou A, Marsh H, et al (1988) Skeletal muscle glycolysis, oxidation, and storage of an oral glucose load. J Clin Invest 81: 1563–1571
Jackson RA, Roshania RD, Hawa MI, Sim BM, DiSilvio L (1986) Impact of glucose ingestion on hepatic and peripheral glucose metabolism in man: an analysis based on simultaneous use of the forearm and double isotope technique. J Clin Endocrinol Metab 63: 541–549
Devos P, Hers H (1979) A molecular order in the synthesis and degradation of glycogen in the liver. Eur J Biochem 99: 161–167
Magnusson I, Schumann W, Bartsch G, et al (1991) Noninvasive tracing of Krebs cycle metabolism in liver. J Biochem 266: 6975–6984
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pimenta, W., Nurjhan, N., Jansson, P.A. et al. Glycogen: its mode of formation and contribution to hepatic glucose output in postabsorptive humans. Diabetologia 37, 697–702 (1994). https://doi.org/10.1007/BF00417694
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00417694