Summary
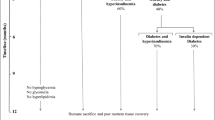
The present study was undertaken to examine the cross-sectional and short-term longitudinal changes in glucose and insulin concentrations as well as measure the enzymatic activity of PEPCK and glycogen synthase in our Psammomys obesus colony. In the cross-sectional study, blood samples were taken from one group of animals at 19 weeks of age (n=37) in the fed state and following a 4-h fast. In a separate group of 19-week-old animals (n = 69), samples were taken l h following an OGTT (1 g/kg body weight) in Psammomys subjected to a 16-h fast. In the longitudinal study, blood samples were taken from one group of animals in the fed state at 7, 11, 15 and 19 weeks of age. All of the cross-sectional data have described the classic inverted U-shaped curve (Starling's curve of the pancreas) in the relationship between glucose and insulin levels. This trend was also reflected by Psammomys subjected to the OGTT; a mild impairment in glucose tolerance was associated with an increase in the insulin response and a further impairment in glucose tolerance was associated with a reduction in the insulin response. Similar results were obtained following a 4-h fast. The short-term longitudinal glucose and insulin data revealed that of the 37 animals examined over the 12-week period, 16 progressed along the inverted U-shaped curve described by the cross-sectional data. Of the other animals, 8 remained unchanged, 7 were unclassifiable and 6 hyperglycaemic Psammomys developed normoglycaemia at the expense of elevated insulin levels. Psammomys were divided into four separate groups based on their glucose and insulin levels in the fed state at age 19 weeks: group A were normoglycaemic (4.5 ± 0.2 mmol/l) and normoinsulinaemic (81±15 mU/l); group B were normoglycaemic (5.3±0.3 mmol/l) and hyperinsulinaemic (267±19 mU/l); group C were hyperglycaemic (16.9±1.4 mmol/l) and hyperinsulinaemic (356±29 mU/l); group D were hyperglycaemic (20.4±1.4 mmol/l) and normoinsulinaemic (92±56 mU/l). Analysis of liver and soleus muscle tissue revealed that active glycogen synthase activity was reduced in the hyperglycaemic Psammomys (C and D). Total glycogen synthase activity (in liver and muscle) and PEPCK activity (in liver) were not different between groups A-D. These findings add to those already published which suggest Psammomys is an ideal animal model for the study of NIDDM.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- NIDDM:
-
Non-insulin-dependent diabetes mellitus
- OGTT:
-
oral glucose tolerance test
- PEPCK:
-
phosphoenolpyruvate carboxykinase
References
Schmidt-Nielsen K, Haines KB (1964) Diabetes mellitus in the sand rat induced by standard laboratory diets. Science 143: 689–690
Hackel DB, Schmidt-Nielsen K, Haines H, Mikat E (1965) Diabetes mellitus in the sand rat (Psammomys obesus): pathologic studies. Lab Invest 14: 200
Kalderon B, Gutman A, Levy E, Shafrir E, Adler JH (1986) Characterization of stages in development of obesitydiabetes syndrome in sand rat (Psammomys obesus). Diabetes 6: 717–724
Adler JH, Lazarovici G, Marton M et al. (1988) Patterns of hyperglycemia, hyperinsulinemia and pancreatic insufficiency in sand rats (Psammomys obesus). In: Shafrir E, Renold AE (eds) Frontiers in diabetes research. Lessons from animal diabetes II. pp 384–388
Prange HD, Schmidt-Nielsen K, Hackel DB (1968) Care and breeding of the fat sand rat (Psammomys obesus Cretzschmar). Lab Anim Care 18: 170–181
Poiley SM (1960) A systematic method of breeder rotation for non-inbred laboratory animal colonies. Proceedings of the Animal Care Panel 10: 159–166
National Health and Medical Research Council (1990) Australian code of practice for the care and use of animals for scientific purposes. Australian Government Publishing Service
Chang HC, Lane MD (1966) The enzymatic carboxylation of phosphoenolpyruvate. J Biol Chem 211: 2413–2420
Golden S, Wals PA, Katz J (1977) An improved procedure for the assay of glycogen synthase and glycogen phosphorylase in rat liver homogenates. Anal Biochem 77: 436–445
Kruszynska YT, Home PD, Alberti KGMM (1986) In vivo regulation of liver and skeletal muscle glycogen synthase activity by glucose and insulin. Diabetes 35: 662–667
Reaven G, Miller R (1968) Study of the relationship between glucose and insulin responses to an oral glucose load in man. Diabetes 70: 560–569
Zimmet P, Whitehouse S, Kiss J (1979) Ethnic variability in the plasma insulin response to oral glucose in Polynesian and Micronesian subjects. Diabetes 28: 624–628
DeFronzo RA, Ferrannini F, Simonson DC (1989) Fasting hyperglycemia in non-insulin-dependent diabetes mellitus: contributions of excessive hepatic glucose production and impaired tissue glucose uptake. Metabolism 38: 387–395
De Fronzo RA (1988) The triumvirate: B cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37: 667–687
Bodkin NL, Metzger BL, Hansen BC (1989) Hepatic glucose production and insulin sensitivity preceding diabetes in monkeys. Am J Physiol 256: E676-E681
Shafrir E (1989) Diabetes in animals. In: H. Rifkin, D. Porte (eds) Diabetes mellitus. pp. 299–340
Collier GR, Greenberg GR, Wolever TMS, Jenkins DJA (1988) The acute effect of fat on insulin secretion. J Clin End Metab 66: 323–326
Reaven GM (1993) Role of insulin resistance in human diseaes (syndrome X): an expanded definition. Annu Rev Med 44: 121–131
Rizza RA, Mandarino LJ, Genest J, Baker BA, Gerich JE (1985) Production of insulin resistance in hyperinsulinemia in man. Diabetologia 28: 70–75
Zimmet PZ, Collins VR, Dowse GK, Knight LT (1992) Hyperinsulinaemia in youth is a predictor for type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 35: 534–541
Arnoff SL, Bennett PH, Gordon P, Rushforth N, Miller M (1977) Unexplained hyperinsulinemia in normal and ‘prediabetic’ Pima Indians compared with normal Caucasians. An example of racial differences in insulin secretion. Diabetes 26: 827–840
Leahy JL (1990) Natural history of B-cell dysfunction in NIDDM. Diabetes Care 13: 992–1010
Shulman GI, Rothman DL, Jue T, Stein P, De Fronzo RA, Shulman RG (1990) Quantitation of muscle glycogen synthesis in normal subjects, and subjects with non-insulin-dependent diabetes by 13C nuclear magnetic resonance spectroscopy. New Eng J Med 322: 223–228
Groop LC, Kankuri M, Schalin-Jantti C et al. (1993) Association between polymorphism of the glycogen synthase gene and non-insulin-dependent diabetes mellitus. New Eng J Med 328: 10–14
Hansen BC, Bodkin NL (1986) Heterogeneity of insulin responses: phases leading to type 2 (non-insulin-dependent) diabetes mellitus in the rhesus monkey. Diabetologia 29: 713–719
Saad MF, Pettitt DJ, Mott DM, Knowler WC, Nelson RG, Bennett PH (1989) Sequential changes in serum insulin concentration during development of non-insulin-dependent diabetes. Lancet I: 356–1358
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barnett, M., Collier, G.R., Collier, F.M. et al. A cross-sectional and short-term longitudinal characterisation of NIDDM in Psammomys obesus. Diabetologia 37, 671–676 (1994). https://doi.org/10.1007/BF00417690
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00417690