Summary
To clarify the contribution of ADCC and NK activities to host immune response against cancer, the characteristics of cells mediating these activities were examined in the peripheral blood lymphocytes of normal volunteers, and the changes of these activities were also evaluated in patients with lung cancer and metastatic pulmonary tumors before and after chemotherapy. OAT cells derived from small cell carcinoma of the lung and K-562 cells derived from erythroleukemia were used as target cells of ADCC and/or NK assay. ADCC and NK activities were not changed according to age, sex, and blood type. Mild and marked personal difference were observed in ADCC and NK activity, respectively. These activities were also influenced by environment. ADCC and NK activities of normal adult volunteers were diversely correlated at the coefficient of γ-0.426.
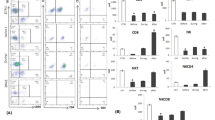
NK activities were high against K-562 and CCRF-CEM cells, and low against BALL and OAT cells. NK activity against K-562 cells was strongly inhibited by K-562 or CCRF-CEM cells with high NK sensitivity, on the other hand, it was slightly inhibited by OAT and BALL cells with low NK sensitivity. NK activity against OAT cells was strongly inhibited by OAT, K-562 and CCRF-CEM cells, but not inhibited by BALL cells. The effector cells mediating NK activity were identified as non-adherent, E-receptor-positive, Fc-receptor-positive small lymphocytes.
NK activity was not decreased before chemotherapy in patients with stage III primary lung cancer and metastatic pulmonary tumors. It was decreased only in patients of bad performance status, and it was significantly decreased in all patients after chemotherapy. ADCC also exhibited the tendency to decrease after chemotherapy in tumor-bearing patients. The recovery of NK-activity after chemotherapy well correlated with the effect of chemotherapy.
Similar content being viewed by others
References
Balch CM, Ades EW, Sohen MR, Shore SL (1980) Human null cells mediating antibody-dependent cellular cytotoxicity express T-lymphocyte differentiation antigens. J Immunol 124:1845–1851
Becker S, Kiessling R, Lee N, Klein G (1978) Modulation of sensitivity to natural killer cell lysis after in vitro explantation of a mouse lymphoma. J Natl Cancer Inst 61:1495–1498
Bolhuis RLH, Schuit HRE, Nooyen AM, Ronteltap CPM (1978) Characterization of natural killer cells and killer cells in human blood: Discrimination between NK and K cell activities. Eur J Immunol 8:731–740
Burton RC, Grail D, Warner NL (1978) Natural cytotoxicity of hemopoietic cell populations against murine lymphoid tumors. Br J Cancer 37:806–817
Callewaert DM, Kaplan J, Johnson DF, Peterson WD (1979) Spontaneous cytotoxicity of cultured human cell lines mediated by normal peripheral blood lymphocytes. Cell Immunol 42:103–112
Callewaert DM, Lightbody JJ, Kaplan J, Jaroszewski J, Peterson WD, Rosenberg JC (1978) Cytotoxicity of human peripheral lymphocytes in cell-mediated lympholysis: Antibody-dependent cell-mediated lympholysis and natural cytotoxicity assays after mixed lymphocyte culture. J Immunol 121:81–85
Droller MJ, Borg H, Perlmann P (1979) In vitro enhancement of natural and antibody-dependent lymphocyte-mediated cytotoxicity against tumor target cells by interferon. Cell Immunol 47:248–260
Eremin O, Ashby J, Stephens JP (1978) Human natural cytoxicity in the blood and lymphoid organs of healthy donors and patients with malignant disease. Int J Cancer 21:35–41
Fujibayashi T, Sato O, Itoh H (1979) Stage-related cell-mediated cytotoxicity with effector cell analysis and comparison of cytotoxic activity of T cells and non-T cells. Cell Immunol 44:225–241
Gupta S, Ferandes G, Nair M, Good R (1978) Spontaneous and antibody-dependent cell-mediated cytotoxicity of human T cell subpopulations. Proc Natl Acad Sci 75:5137–5147
Haliotis T, Roder J, Klein M, Ortaldo J, Fauci AS, Herberman RB (1980) Chediak-Higashi gene in humans I. Impairment of natural-killer function. J Exp Med 151:1039–1048
Herberman RB, Holden HT (1979) Natural killer cells as antitumor effector cells. J Natl Cancer Inst 62:441–445
Hersey P, Edwards A, Honeyman M, McCarthy WH (1979a) Low-natural-killer-cell activity in familial melanoma patients and their relatives. Br J Cancer 40:113–122
Hersey P, Edwards A, Tribivas C, Show H, Milton GW (1979b) Relationship of natural-killer-cell activity to rhesus antigens in man. Br J Cancer 39:234–240
Hochman PS, Cudkowicz G (1979) Suppression of natural cytotoxicity by spleen cells of hydrocortisone-treated mice. J Immunol 123:968–976
Hochman PS, Cudkowicz G, Dausset J (1978) Decline of natural killer cell activity in sublethally irradiated mice. J Natl Cancer Inst 61:265–268
Kay HD, Fagnani R, Bonnard GD (1979) Cytotoxicity against the K-562 erythroleukemia cell line by human natural killer (NK) cells which do not bear free Fc receptors for IgG. Int J Cancer 24:141–150
Kiessling R, Klein E, Wigzell H (1975) Natural killer cells in the mouse. I. Cytotoxic cells with specificity for Moloney leukemia cells. Specificity and distribution according to genotype. Eur J Immunol 5:122–117
Klein M, Roder J, Haliotis T, Koroc S, Jett JR, Herberman RB, Katz P, Fauci AS (1979) Chediak-Higashi gene in humans. II. The selectivity of the defect in natural-killer and antibody-dependent cell-mediated cytotoxicity function. J Exp Med 151:1059–1070
Kleinerman ES, Zwelling LA, Muchmore AV (1980) Enhancement of naturally occurring human spontaneous monocyte-mediated cytotoxicity by cis-diamine-dichlorplatinum (II). Cancer Res 40:3099–3102
Levin AC, Massey RJ, Deinhardt F (1978) Spontaneous human mononuclear cell cytotoxicity to cultured tumor cells: reproducibility of serial measurements with the use of achromium-51-release microcytotoxicity assay. J Natl Cancer Inst 60:1283–1294
Lotzowa E, McCredie KB (1978) Natural killer cells in mice and men, and their possible biological significance. Cancer Immunol Immunother 4:215–221
Mantovani A, Luini W, Peri G, Vecchi A, Spreafico F (1978) Effect of chemotherapeutic agents on natural cell-mediated cytotoxicity in mice. J Natl Cancer Inst 61:1255–1261
Masucci MG, Masucci G, Klein E, Berthold W (1980) Target selectivity of interferon-induced human killer lymphocytes to their Fc receptor expression. Proc Natl Acad Sci 77:3620–3624
McCredie JA, MacDonald HR, Wood SB (1979) Effect of operation and radiotherapy on antibody-dependent cellular cytotoxicity, Cancer 44:99–105
Menon M, Stefani SS (1978) Lymphocyte-mediated natural cytotoxicity in neoplasia. Oncology 35:63–67
Miller RA, Treves A, Kaplan HS (1979) Cytotoxic T-lymphocyte and spontaneous killer-cell activity against human T- and B-lymphoid cell lines. Cell Immunol 47:46–56
Moore K, Moore M (1979) Systemic and in situ natural killer activity in tumor-bearing rats. Br J Cancer 39:636–646
Moore M, Potter MR (1980) Enhancement of human natural cell-mediated cytotoxicity by interferon. Br J Cancer 41:378–387
Mukkerji B, Flowers A, Rothman L, Nathanson L (1980) Spontaneous in vitro cytotoxicity against autochthonous human melanoma cells. J Immunol 124:412–417
Nunn NE, Herberman RB (1979) Natural cytotoxicity of mouse, rat, and human lymphocytes against heterologous target cells. J Natl Cancer Inst 62:765–771
Ochler JR, Lindsay LR, Nunn ME, Herberman RB (1978a) Natural cell-mediated cytotoxicity in rats. I. Tissue and strain distribution and demonstration of a membrane receptor for the Fc portion of IgG. Int J Cancer 21:204–209
Ochler JR, Lindsay LR, Nunn ME, Holden HT, Herberman RB (1978b) Natural cell-mediated cytotoxicity in rats. II. In vivo augmentation of NK-cell activity. Int J Cancer 21:210–220
Ojo E (1979) Positive correlation between the levels of natural killer cells and in vivo resistance to syngeneic tumor transplantations as influenced by various routes of administration of Corynebacterium parvum bacteria. Cell Immunol 45:182–187
Ojo E, Haller O, Kimura A, Wigzell H (1978) An analysis of conditions allowing Corynebacterium parvum to cause either augmentation or inhibition of natural killer cell activity against tumor cells in mice. Int J Cancer 21:444–452
Potter MR, Moore M (1979) Natural cytotoxic reactivity of human lymphocyte subpopulations. Immunology 37:187–194
Pross HF, Baines MG (1976) Spontaneous human lymphocyte-mediated cytotoxicity against tumor target cells. I. The effect of malignant disease, Int J Cancer 18:593–604
Riccardi C, Puccetti P, Santoni A, Herberman RB (1979) Rapid in vivo assay to mouse natural killer cell activity. J Natl Cancer Inst 63:1041–1045
Saal JG, Riethmuller G, Rieber EP, Hadam M, Ehinger H, Schneider W (1977) Regional BCG-therapy of malignant melanoma: in vitro monitoring of spontaneous cytolytic activity of circulating lymphocytes. Cancer Immunol Immunother 3:27–33
Saijo N (1980) Analyses of the natural killer activities of human peripheral blood lymphocytes from normal volunteers and from patients with primary lung cancer and metastatic pulmonary tumors. J Jap Soc Cancer Ther 15:362–314
Saijo N, Irimajiri N, Ozaki A, Shimizu E, Niitani H (1980) Effect of BCG and ACNU on the cytostatic activity of macrophages in normal and tumor-bearing rats. Br J Cancer 42:162–166
Samloska CP (1979) Linoleic acid inhibition of naturally occurring lymphocytotoxicity of breast-derived cells measured by a chromium-51 release assay. J Natl Cancer Inst 62:1427–1433
Santoni A, Herberman RB, Holden HT (1979a) Correlation between natural and antibody-dependent cell-mediated cytotoxicity against tumor target in the mouse. I. Distribution of the reactivity. J Natl Cancer Inst 62:109–116
Santoni A, Herberman RB, Holden HT (1979b) Correlation between natural and antibody-dependent cell-mediated cytotoxicity against tumor target in the mouse. II. Characterization of the effector cells. J Natl Cancer Inst 63:995–1003
Savary CA, Lotzova E (1978) Suppression of natural killer cell cytotoxicity by splenocytes from Corynebacterium parvum-induced bone marrow-tolerant and infant mice. J Immunol 120–239–243
Stanley KE (1980) Prognostic factors for survival in patients with inoperable lung cancer. J Natl Cancer Inst 65:25–32
Stutman O, Paige CJ, Figarella E (1978) Natural cytotoxic cells against solid tumors in mice. I. Strain and age distribution and target cell susceptibility. J Immunol 121:1819–1826
Takasugi M, Akira D (1979) Role of antibodies in the specificity of natural cell-mediated cytotocity. J Natl Cancer Inst 62:1361–1365
Takasugi M, Hirdiwidjaja M, McAllister R, Peer M (1979) Search for specificity in natural cell-mediated cytotoxicity. J Natl Cancer Inst 61:1299–1304
Targan S, Dorey F (1980) Interferon activation of pre-spontaneous killer cells and alternation in kinetics of lysis of both “Pre-SK” and active SK cells. J Immunol 124:2157–2161
Tracey DE (1979) The requirement for macrophages in the augmentation of natural killer cell activity by B.C.G. J Immunol 123:840–845
Yamamura Y, Ogura T, Hirao F, Yasumoto K, Sawamura K, Hattori S, Hayata Y, Kishimoto S, Yamada K, Niitani H, Masaoka T (1981) Phase I study with cell wall skeleton of Nocardia rubra (N-CWS). Cancer Treat Rep 65:707–709
Zarling JM, Schlais J, Eskra L, Greene JJ, Ts'o POP, Carter WA (1980) Augmentation of human natural killer cell activity by polyinosinic acid-polycytidylic acid and its nontoxic mismatched analogues. J Immunol 124:1852–1857
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Saiio, N., Shimizu, E., Irimajiri, N. et al. Analysis of natural killer activity and antibody-dependent cellular cytotoxicity in healthy volunteers and in patients with primary lung cancer and metastatic pulmonary tumors. J Cancer Res Clin Oncol 102, 195–214 (1982). https://doi.org/10.1007/BF00411340
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00411340