Abstract
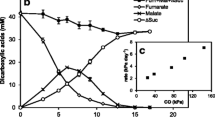
The reduction of elemental or sulfane sulfur to hydrogen sulfide by eubacteria was investigated. Spirillum 5175 had the most active sulfur oxidoreductase. It could be cultivated with fumarate (F), elemental sulfur (S) or nitrate (N) as electron acceptor. Maximum activity was found for Spirillum 5175S but activity was also present in Spirillum 5175F and Spirillum 5175N, i.e. the sulfur oxidoreductase is a constitutive enzyme. It was localized in the membrane, and no activity was found in the cytoplasm in contrast to Desulfovibrio baculatus. Different procedures were applied for the measurement of the sulfur oxidoreductase activity. In the manometric assay hydrogenase was coupled to the sulfur oxidoreductase, and the uptake of dihydrogen was measured in the presence of elemental sulfur. Alternatively, H2S was assayed directly or was trapped in 12% NaOH and determined by the methylene blue procedure. Using 35S sulfur and 35S-labelled compounds both the substrate and H2S could be measured. A further increase in sensitivity was achieved using phenosafranin. It was reduced photochemically, and served as the electron donor to the sulfur oxidoreductase, i.e. no hydrogenase was required. This was an important result in view of the fact that not all sulfur-reducing bacteria contain hydrogenase. However, in those cases the hydrogenase isolated from Clostridium pasteurianum could be coupled to the sulfur oxidoreductase. Among the different forms of elemental sulfur Janek sulfur gave the best results in terms of activity and reproducibility. The reduction of elemental sulfur to hydrogen sulfide had a pH optimum at pH 8.7–8.9. There was always a lag-phase which was pH-dependent. During this period the turbidity of the solution changed. Addition of thiols, such as GSH, shortened the lag-phase and caused an increase in activity of the sulfur oxidoreductase. In the presence of p-chloromercuribenzenesulfonic acid the reaction rate decreased significantly. Comparable reaction rates and activity values of the sulfur oxidoreductase in Spirillum 5175F were obtained with organic trisulfides, RS-S-SR. In contrast to elemental sulfur RS-S-SR are well-defined chemical compounds suitable for quantitative and mechanistic investigations. Labelling the central sulfur of RS-S-SR with 35S gave a satisfactory recovery of the total radioactivity in form of (35S) H2S in our assay. Trisulfides were shown to be formed as reactive intermediates in bacteria. This process required the sulfur transferase rhodanese which was present in Spirillum 5175, or other sulfur-reducing eubacteria.
Similar content being viewed by others
Abbreviations
- EPR:
-
Electron Paramagnetic Resonance
- A:
-
Absorbance
- PCMS:
-
p-chloromercuribenzenesulfonic acid
- Sp. 5175F :
-
Splrillum 5175 grown with fumarate
- Sp. 5175S :
-
with sulfur
- Sp. 5175N :
-
with nitrate
- SCE:
-
Standard Calomel Electrode
References
Bache R (1978) Untersuchungen von c-Typ Cytochromen und eines Eisen-Schwefelproteins aus der Cytoplasmafraktion von Desulfuromonas acetoxidans. Diplomarbeit Univ Göttingen
Bache R, Kroneck PMH, Merkle H, Beinert H (1983) A survey of EPR-detectable components in sulfur-reducing bacteria. Biochim Biophys Acta 722:417–426
Beinert H (1983) Semi-micro methods for analysis of labile sulfide plus sulfane sulfur in unusually stable iron sulfur proteins. Anal Biochem 131:373–378
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cammack R, Fauque G, Moura JJG, Le Gall J (1984) ESR studies of cytochrome c 3 from Desulfovibrio desulfuricans strain Norway4, midpoint potentials of the four haems, and interactions with ferredoxin and colloidal sulphur. Biochim Biophys Acta 784:68–74
Cavallini D, DeMarco C, Mondovi B, Mori BG (1960) The cleavage of cystine by cystathionase and the transulfuration of hypothaurine. Enzymologia 23:161–173
Chen JS, Mortenson LE (1974) Purification and properties of the hydrogenase from Clostridium pasteurianum. Biochim Biophys Acta 371:283–298
Chen JS (1978) Structure and function of two hydrogenases from the dinitrogen-fixing bacterium Clostridium pasteurianum W5. In: Schlegel HG, Schneider K (eds) Hydrogenases: Their catalytic activity, structure and function. Goltze, Göttingen
Fasman GD (ed) (1976) Handbook of Biochemistry and molecular biology. Physical and chemical data, vol I. CRC Press, Cleveland
Fauque G, Herve D, LeGall J (1979) Structure-function relationship in hemoproteins: The role of cytochrome c 3 in the reduction of colloidal sulfur by sulfate-reducing bacteria. Arch Microbiol 121:261–264
Georgi P (1983) Untersuchungen zur Biochemie der Schwefelatmung: Präparation und Physiko-chemische Charakterisierung von Metallproteinen aus Spirillum 5175. Diplomarbeit, Univ Konstanz
Gebhardt NA, Thauer RK, Linder D, Kaulfers PM, Pfennig N (1985) Mechanism of acetate oxidation to CO2 with elemental sulfur in Desulfuromonas acetoxidans. Arch Microbiol 141:392–398
Gornall AG, Bardawill ChJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177:751–766
Hylin JW, Wood JL (1959) Enzymatic formation of polysulfides from mercaptopycuvate. J Biol Chem 234:2141–2144
Janek A (1933) Ein neues Verfahren zur Herstellung von Schwefelsolen. Kolloid Z 664:31–32
Kröger A, Winkler E, Innerhofer A, Hackenberg H, Schägger H (1979) The formate dehydrogenase involved in electron transport from formiate to fumarate in Vibrio succinogenes. Eur J Biochem 94:465–475
Macy JM, Schröder I, Thauer RK, Kröger A (1986) Growth of Wolinella succinogenes on H2S plus fumarate and on formate plus sulfur as energy sources. Arch Microbiol 144:147–150
Massey V, Hemmerich P (1978) Photoreduction of flavoproteins and other biological compounds catalyzed by deazaflavins. Biochemistry 17:9–17
Meyer B (1977) Sulfur, energy and environment. Elsevier Scientific Publishing Company, Amsterdam Oxford New York
Pfennig N, Biebl H (1976) Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic sulfur-reducing, acetate-oxidizing bacterium. Arch Microbiol 110:115–117
Pfennig N, Biebl H (1981) The dissimilatory sulfur-reducing bacteria. In: Starr MP, Stolp H, Trüper HG, Balows A, Schlegel HG (eds) The prokaryotes. A handbook on habitats, isolation and identification of bacteria. Springer, Berlin Heidelberg New York, pp 941–947
Roy AB, Trudinger PA (1970) The Biochemistry of inorganic compounds of sulfur. Cambridge University Press, Cambridge
Sandy JD, Davis RC, Neuberger A (1975) Control of 5-aminolaevulinate synthetase activity in Rhodopseudomonas sphaeroides. A role for trisulfides. Biochim J 150:245–257
Savige WE, Eager J, MacLaren JA, Roxburgh CM (1964) The S-monoxides of cystine, cystamine and homocystine. Tetrahedron Lett 44:3289–3293
Schedel M, Trüper HG (1980) Anaerobic oxidation of thiosulfate and elemental sulfur in Thiobacillus denitrificans. Arch Microbiol 124:205–210
Schink B, Schlegel HG (1979) The membrane-bound hydrogenase of Alcaligenes eutrophus. I. Solubilization and biochemical properties. Biochim Biophys Acta 567:315–324
Schneider JF, Westley J (1969) Metabolic interrelations of sulfur in proteins, thiosulfate and cystine. J Biol Chem 244:5735–5744
Schöberl A, Gräfje H (1958) Aminocarbonsäuren und Haarkeratin mit unsymmetrisch eingebauten Disulfitbindungen, ein Beitrag zum Problem des Disulfitaustausches. Ann Chem 617:71–88
Sörbo B (1955) On the catalytic effect of blood serum on the reaction between colloidal sulfur and cyanide. Acta Chem Scand 9:1656–1660
Szcepkowski TW, Wood JL (1967) The cystathionase-rhodanese system. Biochim Biophys Acta 139:469–478
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev 41:100–180
Umbreit WW, Burris RH, Stauffer JF (1972) Manometric and biochemical techniques. Burgess Publishing Company, Minneapolis
Weitz E, Gieles K, Singer J, Alt B (1956) Über höhere Polythionsäuren. V. Mitteilung: Über die Polythionat-Natur der hydrophilen Odénschen Schwefelsole. Chem Ber 10:2365–2374
Westley J (1973) Rhodanese. Adv Enzymol 39:327–368
White R (1982) Metabolism of L-(sulfane-34S)thiocysteine by Escherichia coli. Biochemistry 21:4271–4275
Winter M (1978) Taxonomische und wachstumsphysiologische Charakterisierung von schwefelreduzierenden Stämmen einer neuen saprophytischen Campylobacter-Art. Diplomarbeit, Univ Göttingen
Wolfe RS, Pfennig N (1977) Reduction of sulfur by spirillum 5175 and syntrophism with Chlorobium. Appl Environ Microbiol 33:427–433
Zöphel A (1986) Untersuchungen zur mikrobiellen Schwefelatmung: Physikochemische und enzymatische Charakterisierung von schwefelreduzierenden Eu- und Archaebakterien. Konstanzer Dissertationen. Hartung-Gorre, Konstanz
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zöphel, A., Kennedy, M.C., Beinert, H. et al. Investigations on microbial sulfur respiration. Arch. Microbiol. 150, 72–77 (1988). https://doi.org/10.1007/BF00409720
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00409720