Summary
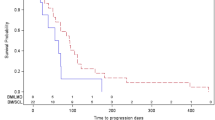
The aim of this study was to examine antitumorigenic Cis DDP properties in metastatic brain tumors. Thirty-four untreated patients with brain metastases recorded by CAT scans or radionuclide scans plus neurological examinations underwent the treatment. Pathohistology of primary tumors mainly showed breast [8] and lung [8] carcinomas and melanomas [10]. Other localizations of primary tumors were infrequent. Cis DDP was administered in doses of 30 mg/m2 body surface daily over 4 days. All the patients received at least two cycles and 33 have been evaluated. No corticosteroids were administered concurrently. An objective response (seven complete and seven partial remissions) was observed in 14 out of 33 patients (42%). Six stable disease cases were also noted. A complete response (5–14 months) was observed in breast cancer [4], lung cancer [1], and melanoma [2]. Seven partial responses lasted 2–5 months. Antitumorigenic activity of Cis DDP was also noted in extracerebral tumor lesions, especially in breast cancer patients. Toxicity was moderate but tolerable. The results of this study have shown Cis DDP to possess antitumorigenic properties also in patients with metastatic brain tumors, a point that has not been proved so far.
Similar content being viewed by others
References
Belt RJ, Himmelstein KJ, Patton TF, Banister SJ, Sternson LA, Repta AJ (1979) Pharmacokinetics of nonprotein bound platinum species following administration of Cis-Dichlorodiamine platinum (II). Cancer Treat Rep 63:1515–1523
Edwards SM, Levin VA, Wilson CB (1980) Brain tumor chemotherapy: an evaluation of agents in current use for phase II and III trials. Cancer Treat Rep 64:1179–1205
Hildebrand J, Brihaye J, Wagenknecht L, Michel J, Kenis Y (1979) Combination chemotherapy with CCNU, vincristine and methotrexate in primary and metastatic brain tumors. Eur J Cancer 11:585–587
Kahn A, McCullough D, Borts F, Sinks LF (1980) Update on use of Cis platinum in CNS malignancies. ASCO, Abstracts 390
Kolarić K, Roth A (1980) Treatment of metastatic brain tumors with the combination of 1-methyl-1-nitrosourea (MNU) and cyclophosphamide. J Cancer Res Clin Oncol 97:193–198
Kolarić K, Roth A, Jeličić I, Matković A (1981) Preliminary report on antitumorigenic activity of Cis dichlorodiammine platinum in metastatic brain tumors. Tumori 67:483–486
Litterst CL, LeRoy AF, Guarino AM (1979) Disposition and distribution of platinum following prenteral administration of Cis Dichlorodiamine Platinum (II) to animals. Cancer Treat Rep 63:1485–1493
Madajewicz S, West CR, Park HC, Avvelanosa AM, Takita H, Karakousis S (1981) Phase II study-Intraarterial BCNU therapy for metastatic brain tumors. Cancer 47:653–657
Madajewicz S, West CR, Avvelanosa AM, Park HC, Karakousis S, Takita H, Wajsman Z (1981) Phase II study of intraarterial Cis Platinum therapy of metastatic brain tumors. UICC Conference on clinical oncology, Lausanne 1981. (Abstracts)
Pouillart P, Mathe G, Thy T (1976) Treatment of malignant gliomas and brain metastases in adults with a combination of adriamycin VM 26, and CCNU: results of a phase II trial. Cancer 38:1909–1916
Seifert W, Caprioli R, Benjamin R, Hester JP (1979) Energy disperisive X-ray fluorescence determination of platinum in plasma, urine and cerebrospinal fluid of patients administered Cis Dichlorodiamine Platinum (DDP). AACR, Abstracts 168
Vick NA, Khandekar JK, Bigner DD (1977) Chemotherapy of brain tumors the “blood-brain barrier” is not a factor. Arch Neurol 34:523–526
WHO handbook for reporting results of cancer treatment (1980) Antitumor drug. ther Rep 6:31–61
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kolarić, K., Roth, A., Jeličic, I. et al. Phase II clinical trial of cis dichlorodiammine platinum (Cis DDP) in metastatic brain tumors. J Cancer Res Clin Oncol 104, 287–293 (1982). https://doi.org/10.1007/BF00406247
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00406247