Abstract
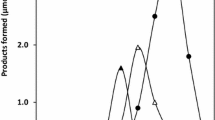
Adenosine deaminase (EC 3.5.4.4) was found to occur in the extract of Azotobacter vinelandii, strain 0, and purified by heating at 65°C, fractionation with ammonium sulfate, DEAE-cellulose chromatography and gel filtration on Sephadex G-150. Purified adenosine deaminase was effectively stabilized by the addition of ethylene glycol. The molecular weight of the enzyme was estimated to be 66,000 by gel filtration on Sephadex G-150. The enzyme specifically attacked adenosine and 2′-deoxyadenosine to the same extent, and formycin A to a lesser extent. The pH optimum of the enzyme was observed at pH 7.2. Double reciprocal plot of initial velocity versus adenosine concentration was concave upward, and Hill interaction coefficient was calculated to be 1.5, suggesting the allosteric binding of the substrate. ATP inhibited adenosine deaminase in an allosteric manner, whereas other nucleotides were without effect. The physiological significance of the enzyme was discussed in relation to salvage pathway of purine nucleotides.
Similar content being viewed by others
References
Andrews, P.: Estimation of the molecular weight of proteins by Sephadex gel filtration. Biochem. J. 91, 222–233 (1964)
Bauer, R. J., Carlberg, D. M.: Adenosine aminohydrolase from Halobacterium cutirubrum. Can. J. Biochem. 51, 621–626 (1973)
Chaney, A. L., Marbach, E. P.: Modified reagents for determination of urea and ammonia. Clin. Chem. 8, 130–132 (1962)
Daddona, P. E., Kelly, W. N.: Human adenosine deaminase. Purification and subunit structure. J. Biol. Chem. 252, 110–115 (1977)
Hoagland, V. D., Jr., Fisher, J. R.: Purification and properties of chicken duodenal adenosine deaminase. J. Biol. Chem. 242, 4343–4351 (1967)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with Folin-phenol reagent. J. Biol. Chem. 193, 265–272 (1951)
Mills, G. C., Schalstieg, F. C., Trimmer, K. B.: Purine metabolism in adenosine deaminase deficiency. Proc. Natl. Acad. Sci. USA 73, 2867–2871 (1976)
Minato, S., Tagawa, T., Naganishi, K.: Studies on nonspecific adenosine deaminase from Takadiastase. I. Purification and properties. J. Biochem. 58, 519–525 (1965)
Nygaard, P.: Adenosine deaminase from Escherichia coli. In: Methods in Enzymology, Vol. 51 (P. A. Hoffee and M. E. Jones, eds.), pp. 508–512. New York: Academic Press 1978
Pickard, M. A.: Purification and some properties of the soluble and membrane-bound adenosine deaminase of Micrococcus sodonensis ATCC 11,880 and their distribution within the family micrococaceae. Can. J. Biochem. 53, 344–353 (1975)
Sakai, T., Jun, H.-K.: Purification and crystallization of adenosine deaminase in Pseudomonas iodinum IFO 3558. FEBS Lett. 86, 174–178 (1978)
Schramm, V. L., Lazorik, F. C.: The pathway of adenylate catabolism in Azotobacter vinelandii. Evidence for adenosine monophosphate nucleosidase as the regulatory enzyme. J. Biol. Chem. 250, 1801–1808 (1975)
Yoshino, M., Ogasawara, N., Suzuki, N., Kotake, Y.: Regulation of AMP nucleosidase in Azotobacter vinelandii. Biochim. Biophys. Acta 146, 620–622 (1967)
Yoshino, M.: AMP nucleosidase from Azotobacter vinelandii. I. Purification and properties. J. Biochem. 68, 321–329 (1970)
Yoshino, M., Tsukada, T., Tsushima, K.: Inosine nucleosidase from Azotobacter vinelandii. Purification and properties. Arch. Microbiol. 119, 59–64 (1978)
Yoshino, M., Tsukada, T., Murakami, K., Tsushima, K.: Adenine nucleotide metabolism in Azotobacter vinelandii. Two metabolic pathways of AMP degradation. Arch. Microbiol. 128, 222–227 (1980)
Zielke, C. L., Suelter, C. H.: Purine purine nucleoside and purine nucleotide aminohydrolases. In: The Enzymes, Vol. 4, 3rd ed. (P. D. Boyer, ed.), pp. 47–78. New York: Academic Press 1971
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tsukada, T., Yoshino, M. Adenosine deaminase from Azotobacter vinelandii. Purification and properties. Arch. Microbiol. 128, 228–232 (1980). https://doi.org/10.1007/BF00406163
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00406163