Summary
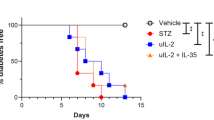
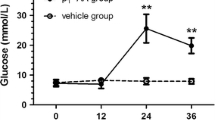
Recent evidence indicates that activated T cells and macrophages play an important role in the induction of insulitis and diabetes in certain strains of mice treated with multiple subdiabetogenic doses of streptozotocin. In the present study, we treated C57BL/6J mice with five daily doses of 40 mg/ml streptozotocin and examined the prophylactic effect of an anti-interleukin 2 receptor monoclonal antibody (PC61). In mice treated with streptozotocin, interleukin 2 receptor-positive mononuclear cells were shown to infiltrate into the islets and soluble interleukin 2 receptors in the sera were significantly increased compared with control mice. The administration of PC61 to the mice attenuated the insulitis, and diminished interleukin 2 receptor-positive cells from islets and soluble interleukin 2 receptors in the sera. Moreover, the administration of PC61 significantly reduced the development of hyperglycaemia shown in these mice (12.8±1.1 mmol/l vs 18.5±0.7 mmol/l, p<0.005). As judged by flow cytometric analysis, this antibody did not cause any changes in either spleen cell counts or T cell subsets. Interleukin 2 receptors were expressed on a minor population of spleen cells regardless of treatment with PC61 (STZ + normal rat IgG: 2.1±0.3%, STZ + PC61: 2.4±0.3%). Even after stimulation of spleen cells with concanavalin A or alloantigen, interleukin 2 receptor expression was not significantly different between the two groups. Our studies suggest that interleukin 2 receptor-positive activated T cells or macrophages are important in the development of multi-low-dose streptozotocin diabetes and that an anti-interleukin 2 receptor antibody can attenuate this process.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Rossini AA, Appel MC, Williams RM, Like AA (1977) Genetic influence of the streptozotocin-induced insulitis and hyperglycemia. Diabetes 26: 916–920
Leiter E (1982) Multiple low-dose streptozotocin-induced hyperglycemia and insulitis in C57BL mice: influence of inbred background, sex, and thymus. Proc Natl Acad Sci USA 79: 630–634
Rossini A, Mordes J, Like A (1985) Immunology of insulin-dependent diabetes mellitus. Annu Rev Immunol 3: 289–320
McEvoy RC, Andersson J, Sandler S, Hellerström C (1984) Multiple low-dose streptozotocin-induced diabetes in the mouse: evidence for stimulation of a cytotoxic cellular immune response against an insulin-producing beta cell line. J Clin Invest 74: 715–722
Rossini A, Like A, Chick W, Appel M, Cahill GL (1977) Studies of streptozotocin-induced insulitis and diabetes. Proc Natl Acad Sci US A 74: 2485–2489
Kiesel U, Kolb H (1983) Suppressive effect of antibodies to immune response gene products on the development of low-dose streptozotocin-induced diabetes. Diabetes 32: 869–871
Rossini AA, Williams RM, Appel MC, Like AA (1978) Complete protection from low-dose streptozotocin-induced diabetes in mice. Nature 276: 182–184
Nedergaard M, Egeberg J, Kromann H (1983) Irradiation protects against pancreatic islet degeneration and hyperglycaemia following streptozotocin treatment of mice. Diabetologia 24: 382–386
Blue M-L, Shin S-I (1984) Diabetes induction by subdiabetogenic doses of streptozotocin in BALB/cBOM mice: noninvolvement of host B-lymphocyte functions. Diabetes 33: 105–110
Kim YT, Steinberg C (1984) Immunologic studies on the induction of diabetes in experimental animals: cellular basis for the induction of diabetes by streptozotocin. Diabetes 33: 771–777
Herold KC, Montag AG, Fitch FW (1987) Treatment with anti-Tlymphocyte antibodies prevents induction of insulitis in mice given multiple doses of streptozotocin. Diabetes 36: 796–801
Bonnevie-Nielsen V, Lernmark Å (1986) An H-2 allo-antiserum preserves β-cell function in mice made diabetic by low-dose streptozotocin. Diabetes 35: 570–573
Kolb-Bachofen V, Epstein S, Kiesel U, Kolb H (1988) Low-dose streptozotocin-induced diabetes in mice: electron microscopy reveals single-cell insulitis before diabetes onset. Diabetes 37: 21–27
Smith KA (1980) T-cell growth factor. Immunol Rev 51: 337–357
Robb RJ, Munck A, Smith KA (1981) T cell growth factor receptors: quantitation, specificity, and biological relevance. J Exp Med 154: 1455–1474
Robb RJ, Green WC, Rusk CM (1984) Low and high affinity cellular receptors for interleukin 2: implication for the level of Tac antigen. J Exp Med 160: 1126–1146
Lowenthal JW, Corthesy P, Tougne C, Lees R, MacDonald HR, Nabholz M (1985) High and low affinity IL-2 receptors: analysis by IL-2 dissociation rate and reactivity with monoclonal anti-receptor antibody PC61. J Immunol 135: 3988–3994
Lowenthal JW, Zubler RH, Nabholz M, MacDonald HR (1985) Similarities between interleukin-2 receptor number and affinity on activated B and T lymphocytes. Nature 315: 669–671
Moreau J-L, Nabholz M, Diamantstein T, Malek T, Shevach E, Thèze J (1987) Monoclonal antibodies identify three epitope clusters on the mouse p55 subunit of the interleukin 2 receptor: relationship to the interleukin 2-binding site. Eur J Immunol 17: 929–935
Malek TR, Robb RJ, Shevach EM (1983) Identification and initial characterization of a rat monoclonal antibody reactive with the murine interleukin 2 receptor-ligand complex. Proc Natl Acad Sci USA 80: 5694–5698
Robb RJ, Mayer PC, Garlick R (1985) Retention of biological activity following radioiodination of human interleukin 2: comparison with biosynthetically labeled growth factor in receptor binding assays. J Immunol Methods 81: 1–4
Robb RJ (1984) Interleukin 2: the molecule and its function. Immunol Today 5: 203–207
Siegel JP, Sharon M, Smith PL, Leonard WJ (1987) The IL-2 receptor β chain (p70): role in mediating signals for LAK, NK and proliferating activities. Science 238: 75–78
Sharon M, Klausner RD, Cullen BR, Chizzonite R, Leonard WJ (1986) Novel interleukin-2 receptor subunit detected by crosslinking under high affinity conditions. Science 234: 859–863
Dukovich M, Wano Y, Thuu LB, Katz P, Cullen BR, Kehrl JH, Green WC (1987) A second human interleukin-2 binding protein that may be a component of high-affinity interleukin-2 receptors. Nature 327: 518–522
Kolb H (1987) Mouse model of insulin dependent-low dose streptozotocin induced diabetes and nonobese diabetic (NOD) mice. Diab Metab Rev 3: 751–778
Oschilewshi M, Schrab E, Kiesel U, Opitz U, Stünkel K, Kolb-Bachofen V, Kolb H (1986) Administration of silica or monoclonal antibody to Thy-1 prevents low-dose streptozotocin-induced diabetes in mice. Immunol Lett 12: 289–294
Cossel L, Schneider E, Kuttler B, Schmidt S, Wohlrab F, Schade J, Bochmann C (1985) Low dose streptozotocin induced diabetes in mice: metabolic light microscopical, histochemical, immuno-fluorescence microscopical, electron microscopical and morphometrical findings. Exp Clin Endocrinol 85: 7–26
Jacobson MP, Rabinovitch A (1986) Spontaneous diabetes mellitus in the BB/W rat: evidence for natural killer cell lysis of islet cells. J Clin Invest 77: 916–924
Jacobson JD, Markmann JF, Brayman KL, Barker CF, Naji A (1988) Prevention of recurrent autoimmune diabetes in BB rats by anti-asialo-GM1 antibody. Diabetes 37: 838–841
Smith KA (1988) Interleukin-2: inception, impact, and implications. Science 240: 1169–1176
Herrmann F, Cannistra SA, Levine H, Griffin JD (1985) Expression of interleukin 2 receptors and binding of interleukin 2 by gamma interferon-induced leukemic and normal monocytic cells. J Exp Med 162: 1111–1116
Rubin LA, Kurman CC, Fritz ME, Biddison WE, Boutin B, Yarchoan R, Nelson DL (1985) Soluble interleukin 2 receptors are released from activated human lymphoid cells in vitro. J Immunol 135: 3172–3177
Keller RJ, Jackson RA (1989) Developmental regulation of serum interleukin-2 receptor concentrations: attenuation of the childhood peak in patients at risk for developing or having recently developed type I diabetes mellitus. J Pediatr 114: 816–819
Giordano C, Panto F, Caruso C, Modica MA, Zambito AM, Sapienza N, Amato MP, Galluzzo A (1989) Interleukin 2 and soluble interleukin 2-receptor secretion defect in vitro in newly diagnosed type I diabetic patients. Diabetes 38: 310–315
Adams DH, Wang L, Hubscher SG, Elias E, Neuberger JM (1989) Soluble interleukin-2 receptors in serum and bile of liver transplant recipients. Lancet 1: 469–471
Manoussukis MN, Papadopoulos GK, Prosos AA, Moutsopoulos HM (1989) Soluble interleukin 2 receptor molecules in the serum of patients with autoimmune disease. Clin Immunol Immunopathol 50: 321–332
Symnous JA, Wood NC, DiGivine FS, Duff GW (1988) Soluble IL-2 receptor in rheumatoid arthritis. J Immunol 141: 2612–2618
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hatamori, N., Yokono, K., Hayakawa, M. et al. Anti-interleukin 2 receptor antibody attenuates low-dose streptozotocin-induced diabetes in mice. Diabetologia 33, 266–271 (1990). https://doi.org/10.1007/BF00403319
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00403319