Abstract
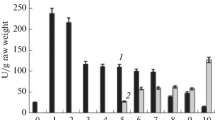
During the greening of etiolated rice leaves, total glutamine synthetase activity increases about twofold, and after 48 h the level of activity usually observed in green leaves is obtained. A density-labeling experiment with deuterium demonstrates that the increase in enzyme activity is due to a synthesis of the enzyme. The enhanced activity obtained upon greening is the result of two different phenomena: there is a fivefold increase of chloroplastic glutamine synthetase content accompanied by a concommitant decrease (twofold) of the cytosolic glutamine synthetase. The increase of chloroplastic glutamine synthetase (GS2) is only inhibited by cycloheximide and not by lincomycin. This result indicates a cytosolic synthesis of GS2. The synthesis of GS2 was confirmed by a quantification of the protein by an immunochemical method. It was demonstrated that GS2 protein content in green leaves is fivefold higher than in etiolated leaves.
Similar content being viewed by others
Abbreviations
- AbH:
-
heavy chain of antibodies
- AbL:
-
light chain of antibodies
- AP:
-
acid phosphatase
- CH:
-
cycloheximide
- G6PDH:
-
glucose-6-phosphate dehydrogenase
- GS:
-
glutamine synthetase
- GS1 :
-
cytosolic glutamine synthetase
- GS2 :
-
chloroplastic glutamine synthetase
- LC:
-
lincomycin
- NAD-MDH:
-
NAD malate dehydrogenase
- NADP-G3PDH:
-
NADP glyceraldehyde-3-phosphate dehydrogenase
References
Bruinsma, J. (1961) A comment on the spectrophotometric determination of chlorophyll. Biochim. Biophys. Acta. 52, 576–578
Ellis, R.J. (1969) Chloroplast ribosome: stereospecificity of inhibition by chloramphenicol. Science 163, 477–478
Gadal, P., Vidal, J., Bouville, J., Godbillon, G. (1981) Influence of light on phosphoenol pyruvate carboxylase in sorghum leaves: immunochemical study, vol. 4, pp. 81–90. Proceedings of the 5th International Photosynthesis Congress. Balaban International Services, Philadelphia
Guitz, C., Hirel, B., Shedlofsky, G., Gadal, P. (1979) Occurrence and influence of light on the relative proportion of two glutamine synthetases in rice leaves. Plant. Sci. Lett. 15, 271–277
Heber, V., Pon, N.G., Heber, M. (1963) Localization of carboxydismutase and triosephosphate dehydrogenase in chloroplasts. Plant Physiol. 38, 355–360
Hirel, B., Gadal, P. (1980a) Glutamine synthetase in rice. A comparative study of the enzymes from roots and leaves. Plant Physiol. 66, 619–623
Hirel, B., Gadal, P. (1980b) Sur la localisation intracellulaire de deux isoformes de la glutamine synthétase dans les feuilles de riz. C.R. Acad. Sci. (Paris) 291, 41–44
Hirel, B., Gadal, P. (1981a) Glutamine synthetase isoforms in pea leaves. Intracellular localization. Z. Pflanzenphysiol. 102, 315–319
Hirel, B., Gadal, P. (1982) Glutamine synthetase isoforms in leaves of a C4 plant: Sorghum vulgare L. Physiol. Plant. 54, 69–74
Hirel, B., Perrot-Rechenmann, C., Suzuki, A., Vidal, J., Gadal, P. (1981c) Glutamine synthetase in spinach leaves. Immunological studies and immunocytochemical localization. Plant Physiol. (in press)
Ifft, J.B., Voet, D.H., Vinograd, J. (1961) The determination of density distribution and density gradients in binary solutions at equilibrium in the ultracentrifuge. J. Phys. Chem. 65, 1138–1145
Ireland, D.M.M., Bradbeer, J.W. (1971) Plastid development in primary leaves of Phaseolus vulgaris. Planta 96, 254–261
Johnson, C.B., Attridge, J.H., Smith, H. (1973) Advantages of the fixed angle rotor for the separation of density labeled form unlabeled proteins by isopicnic equilibrium centrifugation. Biochim. Biophys. Acta 317, 219–230
Kretovich, W.L., Evstigneeva, Z.G., Pushkin, A.V., Dzhokharidze, T.Z. (1981) Two forms of glutamine synthetase in leaves of Cucurbita pepo. Phytochemistry 20, 625–629
Liu, K.D.F., Hugan, A.H.G. (1976) Developmental studies of NAD malate dehydrogenase isozymes in thecotyledon of cucumber seedlings grown in darkness and in light. Planta 131, 279–284
Loiseaux, S. (1976) Ultrastructural modifications of plastids after action of chloramphenicol, lincomycin and rifampicin. Physiol. Vég. 14, 1–10
Malhotra, S.S., Solomos, T., Spencer, M. (1973) Effects of cycloheximide and D-threo chloramphenicol, erythromycin and actinomycine D on de novo synthesis of cytoplasmic and mitochondrial protiens in the cotyledons of germinating pea seeds. Planta 114, 169–184
Mann, A.F., Fentem, P.A., Stewart, G.R. (1979) Identification of two forms of glutamine synthetase in barley (Hordeum vulgare). Biochem. Biophys. Res. Commun. 88, 515–521
Mann, A.F., Fentem, P.A., Stewart, G.R. (1980) Tissue localization in barley (Hordeum vulgare) glutamine synthetase isozymes. FEBS Lett. 110, 265–267
Margulies, M.M. (1964) Effect of chloramphenicol on light dependent synthesis of proteins and enzymes of leaves and chloroplasts of Phaseolus vulgaris. Plant Physiol. 39, 579
Muto, S., Uritani, I. (1970) Glucose-6-phosphate dehydrogenase from sweet potato. Plant Cell Physiol. 11, 767–776
Reger, B.J., Smillie, R.M., Fuller, R.C. (1972) Protein synthesis by the isolated etioplasts and chloroplasts from pea and wheat and the effect of chloramphenicol and cycloheximide. Plant Physiol. 50, 19–23
Scopes, R.K. (1973) Measurement of protein by spectrophotmetry at 205 nm. Anal. Biochem. 59, 277–282
Vidal, J., Rio, M.C., Gadal, P. (1977) Etude de l'évolution de la malate déshydrogénase à NADP durant le verdissement des feuilles de Phaseolus vulgaris L. Plant Sci. Lett. 8, 243–249
Weber, K., Osborn, M. (1969) The reliability for molecular weight determination by dodecyl sulfate-polyacrylamide gel electrophoresis. J. Biol. Chem. 244, 4406–4412
Worthington Enzyme Manual (1972) Worthington biochemical corporation Freehold, New Jersey, 75–76
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hirel, B., Vidal, J. & Gadal, P. Evidence for a cytosolic-dependent light induction of chloroplastic glutamine synthetase during greening of etiolated rice leaves. Planta 155, 17–23 (1982). https://doi.org/10.1007/BF00402926
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00402926