Summary
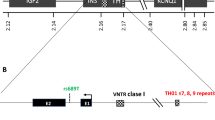
The purpose of this study was to evaluate the role of potential glucokinase defects contributing to susceptibility to Type 2 (non-insulin-dependent) diabetes mellitus in Welsh Caucasians. For this analysis, two microsatellite repeat polymorphisms flanking opposite ends of the gene were employed. For a recently described microsatellite (GCK2), located 6 kilobases upstream of islet exon 1, six different sized alleles were observed, with heterozygosity of 0.50 and polymorphism information content 0.44. Combined heterozygosity with another microsatellite repeat (GCK1) was 0.72. Significant linkage disequilibrium was noted between GCK2 and GCK1, suggesting that haplotypes may be a better predictor of Type 2 diabetes than analysis with either microsatellite alone. Using these two markers, the association with Type 2 diabetes was examined. The frequencies of alleles and genotypes at GCK1 did not differ between the patients with Type 2 diabetes (n=157) and control subjects (n=73). Similarly no differences were observed in GCK2 alleles or genotypes. The frequencies of haplotypes, derived from the two markers, also did not differ between the two groups. To investigate the possibility of minor metabolic effects of glucokinase defects, we also studied the association between the GCK alleles or haplotypes and the response profiles to meal tolerance tests. No association was observed between plasma glucose or insulin responses to meal tolerance tests with GCK haplotypes or alleles. These results suggest that glucokinase mutations in Welsh Caucasians are not major determinants of susceptibility to the common type of Type 2 diabetes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Iynejian P, Girard J (1991) Nomenclature for mammalian glucokinase. Biochem J 275: 821–823
Colowick SP (1973) The hexokinases. In: Boyer PD (ed) The enzymes, vol 9. Academic Press, New York, pp 1–48
Matschinsky FM (1990) Glucokinase as glucose sensor and metabolic signal generator in pancreatic Β-cells and hepatocytes. Diabetes 39: 647–652
Andreone TL, Printz RL, Pilkis SJ, Magnuson MA, Granner DK (1980) The amino acid sequence of rat liver glucokinase deduced from cloned cDNA. J Biol Chem 264: 363–369
Magnuson MA, Shelton KD (1989) An alternate promoter in the glucokinase gene is active in the pancreatic Β cell. J Biol Chem 264: 15936–15942
Magnuson MA, Andreone TL, Printz RL, Koch S, Granner DK (1989) Rat glucokinase gene: structure and regulation by insulin. Proc Natl Acad Sci USA 86: 4838–4842
Tanizawa Y, Koranyi LI, Welling CM, Permutt MA (1991) Human liver glucokinase gene: cloning and sequence determination of two alternatively spliced cDNAs. Proc Natl Acad Sci USA 88: 7294–7297
Koranyi LI, Tanizawa Y, Welling CM, Rabin DU, Permutt MA (1982) Human islet glucokinase gene: isolation and sequence analysis of a full length cDNA. Diabetes 41: 807–811
Matsutani A, Janssen R, Donis-Keller H, Permutt MA (1992) A polymorphic (CA)n repeat element maps the human glucokinase GCK (E.C.2.7.1.1) gene to chromosome 7p. Genomics 12: 319–325
Tanizawa Y, Matsutani A, Chiu KC, Permutt MA (1992) Glucokinase gene: isolation, structural characterization and identification of a microsatellite polymorphism. Molec Endocrinol 6: 1070–1081
Nishi S, Stoffel M, Xiang K, Bell GI, Takeda J (1992) Human pancreatic beta-cell glucokinase gene: cDNA sequence and localization of the polymorphic gene to chromosome 7, band p13. Diabetologia 35: 743–747
Stoffel M, Froguel Ph, Takeda J et al. (1992) Human glucokinase gene: isolation, characterization, and identification of two missense mutations linked to early-onset non-insulin-dependent (type 2) diabetes mellitus. Proc Natl Acad Sci USA 89: 7698–7702
Froguel Ph, Vaxillaire M, Sun F et al. (1992) The glucokinase locus on chromosome 7p is closely linked to early onset non-insulin-dependent diabetes mellitus. Nature 356: 162–164
Hattersley AT, Turner RC, Permutt MA et al. (1992) Type 2 diabetes is linked to the glucokinase gene in a large pedigree. Lancet 339: 1307–1310
Vionnet N, Stoffel M, Takeda J et al. (1992) Nonsense mutation in the glucokinase gene causes early-onset non-insulin dependent diabetes mellitus. Nature 356: 721–722
Stoffel M, Patel P, Lo YMD et al. (1992) Missense mutation in maturity-onset diabetes of the young and mutation screening in late-onset diabetes. Nature Genetics 2: 153–156
Froguel Ph, Vionnet N, Stoffel M et al. (1992) Different phenotypic expression by three mutant alleles of glucokinase gene in MODY. Diabetologia 35 [Suppl 1]: 124A (Abstract)
Velho G, Froguel Ph, Clement K et al. (1992) Primary pancreatic beta-cell secretory defect caused by mutations in glucokinase gene in kindreds of maturity onset diabetes of the young. Lancet 340: 444–448
O'Rahilly, S, Wainscoat JS, Turner RC (1988) Type 2 (non-insulin dependent) diabetes mellitus: new genetics for old nightmares. Diabetes 31: 407–414
Lander ES, Botstein D (1986) Strategies for studying heterogeneous genetic traits in humans by using a linkage map of restriction fragment length polymorphisms. Proc Natl Acad Sci USA 83: 7353–7357
Cox NJ, Bell GI (1989) Disease associations: chance, artifact, or susceptibility genes? Diabetes 38: 947–950
Chiu KC, Province MA, Permutt MA (1992) The glucokinase gene is a genetic marker for non-insulin dependent diabetes mellitus in American Blacks. Diabetes 41: 843–849
Chiu KC, Province MA, Dowse GK et al. (1992) A genetic marker at the glucokinase gene locus for non-insulin-dependent diabetes mellitus in Mauritian Creoles. Diabetologia 35: 632–638
Morgan R, Bishop A, Owens DR, Luzio SD, Peters JR, Rees A (1990) Allelic variants at insulin-receptor and insulin gene loci and susceptibility to NIDDM in Welsh population. Diabetes 39: 1479–1484
Choi W, O'Rahilly S, Buse JB et al. (1991) Molecular scanning of insulin-responsive glucose transporter (Glut4) gene in NIDDM subjects. Diabetes 40: 1712–1718
Grizzle JE, Starmer CF, Koch GG (1969) Analysis of categorical data by linear models. Biometrics 25: 489–504
Orita M, Suzuki Y, Sekiya T, Hayashi K (1989) Rapid and sensitive detection of point mutations and DNA polymorphisms using the polymerase chain reaction. Genomics 5: 874–879
Cox NJ, Xiang K-S, Fajans SS, Bell GI (1992) Mapping diabetes-susceptibility genes. Lessons learned from search for DNA marker for maturity-onset diabetes of the young. Diabetes 41: 401–407
Bodmer WF (1986) Human genetics: the molecular challenge. In: Cold Spring Harbor symposia on quantitative biology, vol L1: molecular biology of homo sapiens. Cold Spring Harbor Laboratory Press, New York, pp 1–13
Cook JTE, Hattersley AT, Christopher P et al. (1992) Linkage analysis of glucokinase gene with NIDDM in caucasian pedigrees. Diabetes 41: 1496–1500
Elbein SC, Hoffman M, Chiu KC, Tanizawa Y, Permutt MA (1993) Linkage analysis of the glucokinase locus in familial type 2 (non-insulin-dependent) diabetic pedigrees. Diabetologia 36: 141–145
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tanizawa, Y., Chiu, K.C., Province, M.A. et al. Two microsatellite repeat polymorphisms flanking opposite ends of the human glucokinase gene: use in haplotype analysis of Welsh Caucasians with Type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 36, 409–413 (1993). https://doi.org/10.1007/BF00402276
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00402276