Abstract
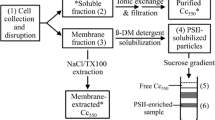
A soluble cytochrome, cytochrome c-551 was purified from an aerobic photosynthetic bacterium Erythrobacter species strain OCh 114 (ATCC No. 33942) by ammonium sulfate fractionation, ion-exchange chromatography and gel-filtration. The cytochrome had absorption maxima at 277, 410, and 524–525 nm in the oxidized form, and at 415, 522, and 550.5 nm in the reduced form. At 77 K, the α-band of the absorption spectrum of the reduced form split in two at 547 and 549 nm. The millimolar absorption coefficient at 550.5 nm was 26.8 mM-1 cm-1 in the reduced form. This cytochrome was an acidic protein with an isoelectric point of 4.9. Its molecular weight was determined to be 15,000 by gel-filtration on Sephadex G-100 and 14,500 by sodium dodecyl sulfate polyacrylamide gel electrophoresis. The midpoint potential of this cytochrome was +250 mV at pH 7.0. This cytochrome did not bind CO.
Similar content being viewed by others
References
Chua NH (1980) Electrophoretic analysis of chloroplast proteins. In: San Pietro A (ed) Methods in Enzymology, vol 69. Academic Press, New York, pp 434–446
Dutton PL, Wilson DF, Lee CP (1970) Oxidation-reduction potentials of cytochromes in mitochondria. Biochemistry 9:5077–5082
Falk JE (1964) Porphyrins and metalloporphyrins. Elsevier, Amsterdam
Harashima K, Nakagawa M, Murata N (1982) Photochemical activities of bacteriochlorophyll in aerobically grown cells of aerobic heterotrophs, Erythrobacter species (OCh 114) and Erythrobacter longus (OCh 101). Plant Cell Physiol 21:185–193
Kimelberg HK, Lee CP (1970) Interaction of cytochrome c with phospholipid membranes II. Reactivity of cytochrome c bound to phospholipid liquid crystals. J Membr Biol 2:252–262
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Matsuura K, Takamiya K, Itoh S, Nishimura M (1980) Effects of surface potential on the equilibrium and kinetics of redox reactions of membrane components with external reagents in chromatophores from Rhodopseudomonas sphaeroides. J Biochem 87:1431–1437
Okamura K, Takamiya K, Nishimura M (1984) Photosynthetic and respiratory electron transfer systems in an aerobic photosynthetic bacterium Erythrobacter sp. strain OCh 114. In: Sybesma C (ed) Advances in photosynthesis research, vol 1. Martinus Nijhoff/Dr. W. Junk, The Hague, pp 641–644
Shiba T, Simidu U, Taga N (1979) Another aerobic bacterium which contains bacteriochlorophyll a. Bull Jap Soc Sci Fish 45:801
Taga N (1968) Some ecological aspects of marine bacteria in the Kuroshio current. Bull Misaki Mar Biol Inst Kyoto Univ 12:56–76
Takamiya K, Hanada H (1980) Cytochrome b 560 in chromatophores from Chromatium vinosum. Plant Cell Physiol 27:979–988
Thomas PE, Ryan D, Levin W (1976) An improved staining procedure for the detection of the peroxidase activity of cytochrome P-450 on sodium dodecyl sulfate polyacrylamide gels. Anal Biochem 75:168–176
Vanderkooi J, Erecińska M, Chance B (1973) Cytochrome c interaction with membranes I. Use of a fluorescent chromophore in the study of cytochrome c interaction with artificial and mitochondrial membranes. Arch Biochem Biophys 154:219–229
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okamura, K., Kisaichi, K., Takamiya, Ki. et al. Purification and some properties of a soluble cytochrome (cytochrome c-551) from Erythrobacter species strain OCh 114. Arch. Microbiol. 139, 143–146 (1984). https://doi.org/10.1007/BF00401989
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00401989