Summary
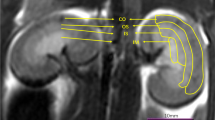
Reductions in the physiological cortical to medullary signal intensity ratio are found in magnetic resonance scans of the kidney in non-diabetic glomerular disease. Whether this abnormality can also characterise patients with Type 1 (insulin-dependent) diabetes mellitus and nephropathy is not known. We measured the cortical to medullary signal intensity ratio in magnetic resonance images of the kidney in 34 patients with Type 1 diabetes (ten with either clinical proteinuria or raised serum creatinine or both, nine with microalbuminuria, seven with normal urinary albumin excretion and long duration of diabetes and eight with Type 1 diabetes of short duration). The cortical to medullary signal intensity ratio showed a trend to cluster at lower values in the normoalbuminuric patients with normal serum creatinine rather than in the nine healthy individuals, independent of Type 1 diabetes duration (1.47 ± 0.06 and 1.41 ± 0.13 vs 1.63 ± 0.16; five groups-Scheffé F-test p = 0.05–0.1). Among the Type 1 diabetic patients, significant reductions in the cortical to medullary signal intensity ratio characterised overt nephropathy (1.19 ± 0.15, p <0.05 vs all groups), but not microalbuminuria (1.47 ± 0.13, p = NS), concomitantly with low glomerular filtration rate and elevated fractional excretion of uric acid, but independent of glycaemic control. The determinants of the renal cortical to medullary signal intensity ratio in Type 1 diabetes are uncertain. Reductions in the cortical to medullary signal intensity ratio may be a late finding in diabetic nephropathy, and parallel the accompanying impairment in kidney haemodynamics. Magnetic resonance imaging of the kidney may not offer clues in the early diagnosis of diabetic nephropathy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Hricak H, Crooks L, Sheldon P, Kaufman L (1983) Nuclear magnetic resonance of the kidney. Radiology 146: 425–432
Leung AW-L, Bydder GM, Steiner RE, Bryant DJ, Young IR (1984) Magnetic resonance imaging of the kidneys. Am J Radiol 143: 1215–1227
Terrier F, Hricak H, Justich E, Dooms GC, Grood W (1986) The diagnostic value of renal cortex-to-medulla contrast on magnetic resonance images. Eur J Radiol 6: 121–126
Marotti M, Hricak H, Terrier F, McAninch JW, Thuroff JW (1987) MR in renal disease: importance of cortical-medullary distinction. Magn Reson Med 5: 160–162
Beomonte Zobel B, Giammarile F, Di Renzi P et al. (1988) Studio della funzionalità renale con risonanza magnetica. Valutazioni Preliminari. Radiol Med 75: 501–504
Otsuji H, Tushima J, Hiroshashi S et al. (1989) Diagnostic value of the cortico-medullary contrast on dynamic CT and MRI in chronic renal parenchymal diseases. (English Abstract). Rinsho Hoshasen 34: 339–343
Lande IM, Glazer GM, Sarnaik S, Aisen A, Rucknagel D, Martel W (1986) Sickle-cell nephropathy: MR imaging. Radiology 158: 379–383
Krolewski AS, Warram JH, Christlieb AR, Busick EJ, Kahn CR (1985) The changing natural history of nephropathy in Type 1 diabetes. Am J Med 78: 785–794
Viberti GC, Bilous RW, Mackintosh D, Keen H (1983) Monitoring glomerular function in diabetic nephropathy. A prospective study. Am J Med 74: 256–264
Viberti GC, Hill RD, Jarrett RJ, Argyropoulos A, Mahmud U, Keen H (1982) Microalbuminuria as a predictor of clinical nephropathy in insulin-dependent diabetes mellitus. Lancet I: 1430–1432
Mathiesen ER, Oxenbøll B, Johansen K, Svendsen PA, Deckert T (1984) Incipient nephropathy in tpye 1 (insulin-dependent) diabetes. Diabetologia 26: 406–410
Mogensen CE, Christensen CK (1984) Predicting diabetic nephropathy in insulin-dependent patients. N Engl J Med 311: 89–93
Parving H-H, Oxenbøll B, Svendsen PA, Christiansen JS, Ander sen AR (1982) Early detection of patients at risk of developing diabetic nephropathy. A longitudinal study of urinary albumin excretion. Acta Endocrinol 100: 550–555
Christensen CK, Mogensen CE (1985) The course of incipient diabetic nephropathy: studies of albumin excretion and blood pressure. Diabetic Med 2: 97–102
Mathiesen ER, Hommel E, Giese J, Parving H-H (1991) Efficacy of captopril in postponing nephropathy in normotensive insulin dependent diabetic patients with microalbuminuria. Br Med J 303: 81–87
Mogensen CE, Chachati A, Christensen CK et al. (1985) Microalbuminuria. An early marker of renal involvement in diabetes. Uremia Invest 9: 85–95
Keen H, Chlouverakis C (1963) An immunoassay for urinary albumin at low concentrations. Lancet II: 913–916
Fabiny DL, Ertinghausen G (1971) Automated reaction rate method for determination of serum creatinine with the Centrifichem. Clin Chem 17: 696–700
Fossati P, Prencipe L, Berti G (1980) Use of 3,5-Dichloro-2-hydroxybenzenesulfonic acid/4-aminophenazone chromogenic system in direct enzymic assay of uric acid in serum and urine. Clin Chem 26: 227–231
Schifreen RS, Hickingbotham JM, Bowers GN (1980) Accuracy, precision and stability in measurement of hemoglobin A 1c by “high performance” cation-exchange chromatography. Clin Chem 26: 466–472
Brøchner-Mortensen J, Giese J, Rossing N (1969) Renal inulin clearance versus total plasma clearance of 51Cr-EDTA. Scand J Clin Lab Invest 23: 301–305
Kaufman L, Crooks LE, Margulis AL (1981) NMR imaging in medicine. Igaku-Shoin, New York, pp 11–52
Herfkens R, Davis P, Crooks L et al. (1981) Nuclear magnetic resonance imaging of the abnormal live rat and correlations with tissue characteristics. Radiology 141: 211–218
Thickman D, Kundel H, Biery D (1984) Magnetic resonance evaluation of hydronephrosis in the dog. Radiology 152: 113–116
Brenner BM (1985) Nephron adaptation to renal injury or ablation. Am J Physiol 249: F324-F337
Skøtt P, Mathiesen ER, Hommel E, Gall M-A, Bruun NE, Parving H-H (1989) The increased proximal tubular reabsorption of sodium and water is maintained in long-term insulin-dependent diabetics with early nephropathy. Scand J Clin Lab Invest 49: 419–425
Semplicini A, Angeli P, Nosadini R et al. (1989) Tubular Na+ handling in Type 1 insulin-dependent diabetics during saline and ketone body infusion. Diabetes Res 11: 67–72
Weidmann P, Ferrari P (1991) Central role of sodium in hypertension in diabetic subjects. Diabetes Care 14: 220–232
Feldt-Rasmussen B, Mathiesen ER, Deckert T et al. (1987) Central role for sodium in the pathogenesis of blood pressure changes independent of angiotensin, aldosterone and catecholamines in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 30: 610–617
Koomans HA, Boer WH, Mees EJD (1989) Evaluation of lithium clearance as a marker of proximal tubule sodium handling. Kidney Int 36: 2–12
Jennerholm S, Backman U, Bohman S-O (1990) Magnetic resonance imaging of the transplanted kidney. Acta Radiol 31: 499–503
Østerby R, Parving H-H, Nyberg G, Hommel E, Mauer SM, Steffes MW (1989) Morphology of diabetic glomerulopathy and relationship to hypertension. Diabete Metabolisme 15: 278–283
Frøkjær-Thomsen O, Andersen AR, Christiansen JS, Deckert T (1984) Renal changes in long-term type 1 (insulin-dependent) diabetic patients with and without clinical nephropathy: a light microscopic, morphometric study of autopsy material. Diabetologia 26: 361–365
Bilous RW, Mauer SM, Sutherland DER, Steffes MW (1989) Mean glomerular volume and rate of development of diabetic nephropathy. Diabetes 38: 1142–1147
Chavers BM, Bilous RW, Ellis EN, Steffes MW, Mauer SM (1989) Glomerular lesions and urinary albumin excretion in type 1 diabetes without proteinuria. N Engl J Med 320: 966–970
Wiseman MJ, Viberti GC (1983) Kidney size and glomerular filtration rate in type 1 (insulin-dependent) diabetes mellitus revisited. Diabetologia 25: 530 (Letter)
Jones SL, Wiseman MJ, Viberti GC (1991) Glomerular hyperfiltration as a risk factor for diabetic nephropathy: five-year report of a prospective study. Diabetologia 34: 59–60 (Letter)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mangili, R., Sironi, S., Rankel, G. et al. Magnetic resonance imaging of the kidney in Type 1 (insulin-dependent) diabetes mellitus. Diabetologia 35, 1002–1008 (1992). https://doi.org/10.1007/BF00401433
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00401433