Summary
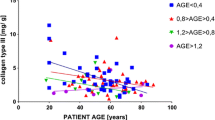
Arterial glycosaminoglycans are considered to be important in atherogenesis due to their ability to trap lipid inside the vessel wall and to influence cellular migration and proliferation. Atherosclerotic lesions have displayed an altered glycosaminoglycan content and distribution. Diabetes is a recognized risk factor for atherosclerosis, but no information is available on the arterial glycosaminoglycans in human diabetes. We examined glycosaminoglycans in normal and atherosclerotic intima of non-diabetic and Type 2 (non-insulin-dependent) diabetic patients. Intima was stripped from autopsy samples of thoracic aortas; normal and plaque areas were separated. Glycosaminoglycans were isolated by delipidation, proteolytic digestion, and precipitation and characterized by quantitation of total glycosaminoglycan and evaluation of glycosaminoglycan distribution by electrophoresis and densitometry. Results indicate a significant decrease in total glycosaminoglycan and significant changes in their distribution in atherosclerotic plaques: a relative decrease in heparan sulphate, a relative increase in dermatan sulphate and thus a decrease in the ratio of heparan sulphate to dermatan sulphate. A similar but less marked change in the ratio was found in normal intima of diabetic subjects, while in their plaques this change was more pronounced. This suggests that changes in arterial glycosaminoglycans (especially the ratio of heparan sulphate to dermatan sulphate) precede the development of lesions in diabetes and may be important in atherogenesis.
Article PDF
Similar content being viewed by others
References
Camejo G (1982) The interaction of lipids and lipoproteins with intercellular matrix of arterial tissue: its possible role in atherogenesis. Adv Lipid Res 19: 1–53
Wight TN (1989) Cell biology of arterial proteoglycans. Arteriosclerosis 9: 1–20
Ruoslahti E (1989) Proteoglycans in cell regulation. J Biol Chem 264: 13369–13372
Buddecke E (1962) Chemical changes in the ground substance of the vessel wall in arteriosclerosis. J Ather Res 2: 32–46
Smith EB (1965) The influence of age and atherosclerosis on the chemistry of aortic intima: Part 2. collagen and mucopolysaccharides. J Ather Res 5: 241–248
Klynstra FB, Bottcher CJF, Van Melsen JA, Van Der Laan EJ (1967) Distribution and composition of acid mucopolysaccharides in normal and atherosclerotic human aortas. J Ather Res 7: 301–303
Kumar V, Berenson GS, Ruiz H, Dalferes ER Jr, Strong JP (1967) Acidic mucopolysaccharides of human aorta: Part 2. Variation with atherosclerotic involvement. J Ather Res 7: 583–590
Dalferes ER Jr, Ruiz H, Kumar V, Radhakrishnamurthy B, Berenson GS (1971) Acid mucopolysaccharides of fatty streaks in young, human male aortas. Atherosclerosis 13: 121–131
Stevens RL, Colombo M, Gonzales JJ, Hollander W, Schmid K (1976) The glycosaminoglycans of the human artery and their changes in atherosclerosis. J Clin Invest 58: 470–481
Tammi M, Seppala PO, Lehtonen A, Mottonen M (1978) Connective tissue components in normal and atherosclerotic human coronary arteries. Atherosclerosis 29: 191–194
Murata K, Yokoyama Y (1982) Acidic glycosaminoglycan, lipid and water contents in human coronary arterial branches. Atherosclerosis 45: 53–65
Yla-Herttuala S, Sumuvuori H, Karkola K, Mottonen M, Nikkari T (1986) Glycosaminoglycans in normal and atherosclerotic human coronary arteries. Lab Invest 54: 402–407
Murata K, Yokoyama Y (1989) Acidic glycosaminoglycans in human atherosclerotic cerebral arterial tissues. Atherosclerosis 78: 69–79
Hollman J, Schmidt A, Bassewitz D, Buddecke E (1989) Relationship of sulfated glycosaminoglycans and cholesterol content in normal and arteriosclerotic human aorta. Arteriosclerosis 9: 154–158
Wagner WD, Salisbury BGJ (1978) Aortic total glycosaminoglycan and dermatan sulfate changes in atherosclerotic Rhesus monkeys. Lab Invest 39: 328–332
Alavi M, Moore S (1985) Glycosaminoglycan composition and biosynthesis in the endothelium-covered neointima of de-endothelialized rabbit aorta. Exp Mol Pathol 42: 389–400
Salisbury BGJ, Hajjar DP, Minick CR (1985) Altered glycosaminoglycan metabolism in injured arterial wall. Exp Mol Pathol 42: 306–319
Robertson WB, Strong JP (1968) Atherosclerosis in persons with hypertension and diabetes mellitus. Lab Invest 18: 538–551
Kannel WB, McGee WL (1978) Diabetes and cardiovascular disease. The Framingham Study. JAMA 241: 2035–2038
Ruderman NB, Haudenschild C (1984) Diabetes as an atherogenic factor. Progress in Cardiovas Dis 26: 373–412
Moore S (1981) Response of the arterial wall to injury. Diabetes 30 [Suppl 2]: 8–13
Moore S (1985) Pathogenesis of atherosclerosis. Metabolism 34 [Suppl 1]: 13–16
Sternberg M, Cohen-Forterre L, Peyroux J (1985) Connective tissue in diabetes mellitus: biochemical alterations of the intercellular matrix with special reference to proteoglycans, collagens and basement membranes. Diabete Metab 11: 27–50
Ichida T, Kalant N (1968) Aortic glycosaminoglycans in atheroma and alloxan diabetes. Can J Biochem 46: 249–260
Cohen MP, Foglia VG (1970) Aortic mucopolysaccharides in experimental diabetes. Diabetes 19: 639–643
Malathy K, Kurup PA (1972) Metabolism of glycosaminoglycans in alloxan diabetic rats. Diabetes 21: 1162–1167
Sirek OV, Sirek A, Cukerman E (1980) Arterial glycosaminoglycans in diabetic dogs. Blood Vessels 17: 271–275
Roden L, Baker JR, Cifonelli A, Mathews MB (1972) Isolation and characterization of connective tissue polysaccharides. Method Enzymol 28: 73–140
Folch J, Lees M, Sloane-Stanley GH (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226: 497–509
Hronowsky L, Anastassiades TP (1979) Quantitation and interaction of glycosaminoglycans with alcian blue in dimethyl sulfoxide solutions. Anal Biochem 93: 60–72
Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acids. Anal Biochem 54: 484–489
Alavi MZ, Moore S (1987) Proteoglycan composition of rabbit arterial wall under conditions of experimentally induced atherosclerosis. Atherosclerosis 63: 65–74
Dalferes ER Jr, Radhakrishnamurthy B, Ruitz HA, Berenson GS (1987) Composition of proteoglycans from human atherosclerotic lesions. Exp Mol Pathol 47: 363–376
Cherchi GM, Coinu R, Demuro P et al. (1990) Structural and functional modifications of human aorta proteoglycans in atherosclerosis. Matrix 10: 362–372
Ying-Shan C, Chung-Ling Z, Pei-Zhen Z, Zhuo-Lin D (1991) Human aortic proteoglycans of subjects from districts of high and low prevalence of atherosclerosis in China. Atherosclerosis 86: 9–15
Alavi MZ, Wasty F, Li Z, Galis ZS, Ismail N, Moore S (1992) Enhanced incorporation of [14C]glucosamine into glycosaminoglycans of aortic neointima of balloon-injured and cholesterolfed rabbits in vitro. Atherosclerosis 95: 59–67
Moore S (1981) Injury mechanisms in atherogenesis. In: Moore S (ed) Vascular injury and atherosclerosis. Marcel Dekker, New York, pp 131–148
Ross R (1986) The pathogenesis of atherosclerosis-an update. New Engl J Med 314: 488–500
Hollman J, Thiel J, Schmidt A, Buddecke E (1985) Increased activity of chondroitin sulfate-synthesizing enzymes during proliferation of arterial smooth muscle cells. Exp Cell Res 167: 484–499
Alavi MZ, Richardson M, Moore S (1989) The in vitro interactions between serum lipoproteins and proteoglycans of the neointima of rabbit aorta after a single balloon catheter injury. Am J Pathol 134: 287–294
Castellot JJ Jr, Karnovsky MJ (1987) Heparin and the regulation of growth in the vascular wall. In: Campbell JH, Campbell GR (eds) Vascular smooth muscle in culture. CRC Press, Boca Raton, pp 93–115
Majack RA, Clowes AW (1984) Inhibition of vascular smooth muscle cell migration by heparin-like glycosaminoglycans. J Cell Physiol 118: 253–256
Breton M, Berrou E, Deudon E, Brahimi-Horn M, Picard J (1988) Effect of insulin on sulfated proteoglycan synthesis in cultured smooth muscle cells from pig aorta. Exp Cell Res 177: 212–220
Dolgov VV, Zaikina OE, Bondarenko MF, Repin VS (1982) Aortic endothelium of alloxan diabetic rabbits: a quantitative study using scanning electron microscopy. Diabetologia 22: 338–343
Hadcock S, Richardson M, Winocour PD, Hatton MWC (1991) Intimal alterations in rabbit aortas during the first 6 months of alloxan-induced diabetes. Arterioscler Thromb 11: 517–529
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wasty, F., Alavi, M.Z. & Moore, S. Distribution of glycosaminoglycans in the intima of human aortas: changes in atherosclerosis and diabetes mellitus. Diabetologia 36, 316–322 (1993). https://doi.org/10.1007/BF00400234
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400234