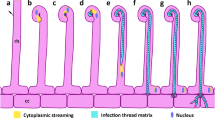
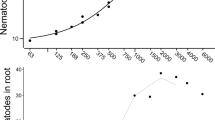
Abstract
In soybean (Glycine max (L.) Merr.) the uninfected cells of the root nodule are responsible for the final steps in ureide production from recently fixed nitrogen. Stereological methods and an original quantitative method were used to investigate the organization of these cells and their spatial relationships to infected cells in the central region of nodules of soybean inoculated with Rhizobium japonicum strain USDA 3I1B110 and grown with and without nitrogen (as nitrate) in the nutrient medium. The volume occupied by the uninfected tissue was 21% of the total volume of the central infected region for nodules of plants grown without nitrate, and 31% for nodules of plants grown with nitrate. Despite their low relative volume, the uninfected cells outnumbered the much larger infected cells in nodules of plants grown both without and with nitrate. The surface density of the interface between the ininfected and infected tissue in the infected region was similar for nodules in both cases also, the total range being from 24 to 26 mm2/mm3. In nodules of plants grown without nitrate, all sampled infected cells were found to be in contact with at least one uninfected cell. The study demonstrates that although the uninfected tissue in soybean nodules occupies a relatively small volume, it is organized so as to produce a large surface area for interaction with the infected tissue.
Similar content being viewed by others
References
Allen, O.N., Allen, E.K. (1940) Response of the peanut plant to inoculation with Rhizobia, with special reference to morphological development of the nodules. Bot. Gaz. 102, 121–142
Arora, N. (1954) Morphological development of the root and stem nodules of Aeschynomene indica L. Phytomorphology 4, 211–216
Bergersen, F.J. (1961) Haemoglobin content of legume root nodules. Biochim. Biophys. Acta 50, 576–578
Bergersen, F.J. (1983) Root nodules of legumes: structure and functions. Research Studies Press/Wiley, New York
Bergersen, F.J., Goodchild, D.J. (1973) Aeration pathways in soybean root nodules. Aust. J. Biol. Sci. 26, 729–740
Berlyn, G.P., Miksche, J.P. (1976) Botanical microtechnique and cytochemistry. Iowa State Univ. Press, Ames, USA
Bieberdorf, F.W. (1938) The cytology and histology of the root nodules of some leguminosae. J. Am. Soc. Agron. 30, 375–389
Bond, L. (1948) Origin and developmental morphology of root nodules of Pisum sativum. Bot. Gaz. 109, 411–434
Chandler, M.R., (1978) Some observations on infection of Arachis hypogaea L. by Rhizobium. J. Exp. Bot. 29, 749–755
Chandler, M.R., Dale, R.A., Roughley, R.J. (1982) Infection and root-nodule development in Stylosanthes species by Rhizobium. J. Exp. Bot. 33, 47–57
Chen, H.K., Thornton, H.G. (1940) The structure of “ineffective” nodules and its influence on nitrogen fixation. Proc. R. Soc. London B 129, 208–229
Dangeard, P.A. (1926) Recherches sur les tubercules radicaux des Légumineuses. Botaniste 16, 1–269
Dart, P.J. (1977) Infection and development of leguminous root nodules. In: A treatise on dinitrogen fixation, vol. 3: Biology, pp. 367–472, Hardy, R.W.F., Silver, W.S. eds. Wiley, New York
Hanks, J.F., Schubert, K.R., Tolbert, N.E. (1983) Isolation and characterization of infected and uninfected cells from soybean nodules. Plant Physiol. 71, 869–873
Harper, J.E., Gibson, A.H. (1984) Differential nodulation tolerance to nitrate among legume species. Crop Sci. 24, 797–801
Herridge, D.F. (1984) Effects of nitrate and plant development on the abundance of nitrogenous solutes in root-bleeding and vacuum-extracted exudates of soybean. Crop Sci. 24, 173–181
Hoagland, D.R., Snyder, W.C. (1933) Nutrition of strawberry plant under controlled conditions. Proc. Am. Soc. Hort. Sci. 30, 288–294
Lechtova-Trnka, M. (1931) Etude sur les bactéries des légumineuses et observations sur quelques champignons parasites des nodosités. Botaniste 23, 301–531
Newcomb, E.H., Tandon, S.R. (1981) Uninfected cells of soybean root nodules: ultrastructure suggests key role in ureide production. Science 212, 1394–1396
Newcomb, E.H., Selker, J.M.L., Tandon, S.R., Meng, F.-J., Kowal, R.R. (1985a) Uninfected cells in ureide-and amideexporting legume root nodules. In: Nitrogen fixation and CO2 metabolism, pp. 31–40, Ludden, P.W., Burris, J.E., eds. Elsevier Science, New York
Newcomb, E.H., Tandon, S.R., Kowal, R.R. (1985b) Ultrastructural specialization for ureide production in uninfected cells of soybean root nodules. Protoplasma 125, 1–12
Newcomb, W. (1981) Nodule morphogenesis and differentiation. Int. Rev. Cytol. Suppl. No. 13, 247–298
Pankhurst, C.E. (1970) Development of effective and ineffective root nodules on two Lotus species. N. Z. J. Sci. 13, 519–531
Pugh, T.D., King, J.H., Koen, H., Nychka, D., Chover, J., Wahba, G., He, Y., Goldfarb, S. (1983) Reliable stereological method for estimating the number of microscopic hepatocellular foci from their transections. Cancer Res. 43, 1261–1268
Ralston, E.J., Imsande, J. (1983) Nodulation of hydroponically grown soybean plants and inhibition of nodule development by nitrate. J. Exp. Bot. 34, 1371–1378
Roughley, R.J. (1970) The influence of root temperature, Rhizobium strain and host selection on the structure and nitrogen-fixing efficiency of the root nodules of Trifolium subteraneum. Ann. Bot. 34, 631–646
Sachs, T. (1978) The development of spacing patterns in the leaf epidermis. In: The clonal basis of development, pp. 161–183, Subtelny, S., Sussex, I.M., eds. 36th Symp. Soc. Devel. Biol. Academic Press, New York London
Sprent, J.I. (1971) The effects of water stress on nitrogen-fixing root nodules. I. Effects on the physiology of detached soybean nodules. New Phytol. 70, 9–17
Spurr, A.R. (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43
Steel, R.G.D., Torrie, J.H. (1960) Principles and procedures of statistics. McGraw-Hill, New York
Toth, R. (1982) An introduction to morphometric cytology and its application to botanical research. Am. J. Bot. 69, 1694–1706
Weibel, E.R. (1979) Stereological methods, vol. I: Practical methods for biological morphometry. Academic Press, New York London
Weibel, E.R., Kistler, G.S., Scherle, W.F. (1966) Practical stereological methods for morphometric cytology. J. Cell Biol. 30, 23–38
Wood, S.M., Layzell, D.B., Newcomb, W., Pankhurst, C.E. (1985) A morphometric study of effective nodules induced by Rhizobium loti and Bradyrhizobium sp. (Lotus) on Lotus pedunculatus. Can. J. Bot. 63, 43–53
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Selker, J.M.L., Newcomb, E.H. Spatial relationships between uninfected and infected cells in root nodules of soybean. Planta 165, 446–454 (1985). https://doi.org/10.1007/BF00398089
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00398089