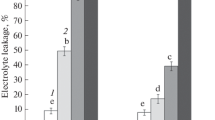
Abstract
The effect of osmotic dehydration on metabolic reactions in three different subcellular compartments (chloroplast, cytoplasm and mitochondria) was studied in vacuum-infiltrated thin leaf slices from various plants, in the absence of stomatal control. The reactions tested were CO2 fixation in the light (chloroplast), CO2 fixation in the dark (cytoplasm), and O2 uptake in the dark (mitochondria). In most plants, the sensitivity of dark CO2 fixation to dehydration was similar to the sensitivity of photosynthesis. In leaf slices from a plant with Crassulacean acid metabolism (Kalanchoe pinnata), dark CO2 fixation (which reached similar rates as light fixation) was slightly more sensitive to osmotic stress than photosynthesis. Dark respiration (measured as O2 uptake) was significantly more resistant to hypertonic stress than both types of CO2 fixation. In crude leaf extracts from spinach, the response of soluble enzymes from the three different subcellular compartments to high concentrations of various electrolytes and neutral compounds was examined and compared with the in-vivo data.
Similar content being viewed by others
Abbreviations
- CAM:
-
Crassulacean acid metabolism
- PEP:
-
phosphoenolpyruvate
- π○ :
-
osmotic potential
References
Arnon, D.J. (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 24, 1–15
Beer, S., Shomer-Ilan, A., Waisel, Y. (1975) Salt-stimulated Phosphoenolpyruvate carboxylase in Cakile maritima. Physiol. Plant. 34, 293–295
Bergmeyer, H.U., Gawehn, K., Graßl, M. (1970) Enzyme I. In: Methoden der enzymatischen Analyse, vol. 1, pp. 411–412, Bergmeyer, H.U., ed. Verlag Chemie, Weinheim
Böcher, M., Kluge, M. (1977) Der C4-Weg der C-Fixierung bei Spinacia oleracea. I. 14C-Markierungsmuster suspendierter Blattstreifen unter dem Einfluß des Suspensionsmediums. Z. Pflanzenphysiol. 83, 347–361
Clarkson, D.T., Hanson, J.B. (1980) The mineral nutrition of higher plants. Annu. Rev. Plant Physiol. 31, 239–298
Coughlan, S.J., Wyn Jones, R.G. (1980) Some responses of Spinacia oleracea to salt stress. J. Exp. Bot. 31, 883–893
Flowers, T.J., Hanson, J.B. (1969) The effect of reduced water potential on soybean mitochondria. Plant Physiol. 44, 939–945
Flowers, T.J., Troke, P.F., Yeo, A.R. (1977) The mechanism of salt tolerance in halophytes. Annu. Rev. Plant Physiol. 28, 89–121
Hanson, A.D., Hitz, W.D. (1982) Metabolic responses of mesophytes to plant water deficits. Annu. Rev. Plant Physiol 33, 163–203
Hsiao, T.C. (1973) Plant responses to water stress. Annu. Rev. Plant Physiol. 24, 519–570
Kaiser, W.M. (1982) Correlation between changes in photosynthetic activity and changes in total protoplast volume in leaf tissue from hygro-, meso- and xerophytes under osmotic stress. Planta 154, 538–545
Kaiser, W.M., Heber, U. (1981) Photosynthesis under osmotic stress. Effect of high solute concentrations on the permeability properties of the chloroplast envelope and on activities of stroma enzymes. Planta 153, 423–429
Kaiser, W.M., Kaiser, G., Prachuab, P.K., Wildman, S.G., Heber, U. (1981a) Photosynthesis under osmotic stress. Inhibition of photosynthesis of intact chloroplasts, protoplasts and leaf slices at high osmotic potentials. Planta 153, 416–422
Kaiser, W.M., Kaiser, G., Schöner, S., Neimanis, S. (1981b) Photosynthesis under osmotic stress. Differential recovery of photosynthetic activities of stroma enzymes, intact chloroplasts, protoplasts and leaf slices after exposure to high solute concentrations. Planta 153, 530–435
Kaiser, W.M., Paul, J., Bassham, J.A. (1979) Release of photosynthates from mesophyll cells in vitro and in vivo. Z. Pflanzenphysiol. 94, 377–385
O'Leary, M.H. (1982) Phosphoenolpyruvate carboxylase: an enzymologists view. Annu. Rev. Plant Physiol. 33, 297–315
Osmond, C.B. (1976) Ion absorption and carbon metabolism in cells of higher plants. In: Encyclopaedia of plant physiology, N.S., vol. 2 A: Transport in plants II, pp. 347–372, Lüttge, U., Pitman, M.G., eds. Springer, Berlin Heidelberg New York
Paleg, L.g., Douglas, T.J., v. Daal, A., Keech, D.B. (1981) Proline, Betaine and other organic solutes protect enzymes against heat inactivation. Aust. J. Plant Physiol. 8, 107–114
Pedersen, T.A., Kirk, M.R., Bassham, J.A. (1966) Light-dark transients in the levels of intermediate compounds during photosynthesis in air-adapted Chlorella. Physiol. Plant. 19, 219–231
Pollard, A., Wyn Jones, R.G. (1979) Enzyme activities in concentrated solutions of glycinebetaine and other solutes. Planta 144, 291–298
Stewart, C.R. (1981) Proline accumulation: biochemical aspects. In: The physiology and biochemistry of drought resistance in plants, pp. 243–259, Paleg, L.G., Aspinall, D., eds. Academic Press, Sydney New York London Toronto San Francisco
Winter, K., Lüttge, U. (1976) Malate accumulation in leaf slices of Mesembryanthemum cristallinum in relation to osmotic gradients between cells and the medium. Aust. J. Plant Physiol. 3, 653–663
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaiser, W.M., Schwitulla, M. & Wirth, E. Reactions in chloroplasts, cytoplasm and mitochondria of leaf slices under osmotic stress. Planta 158, 302–308 (1983). https://doi.org/10.1007/BF00397331
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00397331