Abstract
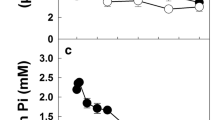
Tonoplast vesicles were prepared from red-beet (Beta vulgaris L. ssp. conditiva) hypocotyl tubers (“beetroot”) known to store sucrose. Uptake experiments, employing uridine 5′-diphospho-[14C]glucose (UDP-[14C]glucose) showed the operation of an UDP-glucose-dependent group translocator for vectorial synthesis and accumulation of sucrose, recently described for sugarcane and red-beet vacuoles and for tonoplast vesicles prepared from sugarcane suspension cells. Characterization of the kinetic properties yielded the following results. Uptake of UDP-glucose was linear for 15 min. The apparent K m was 0.75 mM for UDP-glucose (at pH 7.2, 1 mM Mg2+), V max was 32 nmol·(mg protein)-1·min-1. The incorporation of UDP-glucose exhibited a sigmoidal substrate-saturation curve in the absence of Mg2+, the Hill coefficient (n H) was 1.33; Michaelis-Menten kinetics were obtained, however, in the presence of 1 mM MgCl2. For the reaction sequence under the control of the group translocator a dual pH optimum was found at pH 7.2 and 7.9, respectively. All reaction intermediates and the end product sucrose could be identified by two-dimensional high-performance thin-layer chromatography and autoradiography. The distribution pattern of radioactivity showed almost uniformly high labeling of all intermediates and sucrose. The physiological relevance of the results is discussed in the light of the fact that the tonoplast of red-beet storage cells accommodates two mechanisms of sucrose uptake (i) vectorial sucrose synthesis and (ii) direct ATP-dependent sucrose assimilation.
Similar content being viewed by others
Abbreviations
- HPTLC:
-
High-performance thin-layer chromatography
- UDP:
-
uridine 5′-diphosphate
- SDS:
-
sodium dodecyl sulfate
References
Bennett, A.B., O'Neill, S.D., Spanswick, R.M. (1984) H+-ATPase activity from storage tissue of Beta vulgaris. I. Identification and characterization of an anion-sensitive H+-ATPase. Plant Physiol. 74, 538–544
Bennett, A.B., Spanswick, R.M. (1983) Optical measurements of pH and ΔΨ in corn root membrane vesicles: Kinetic analysis of Cl--effects on a H+-translocating ATPase. J. Membr. Biol 71, 95–107
Briskin, D.P., Poole, R.J. (1983) Characterization of a K+-stimulated ATP-ase associated with the plasma membrane of Red Beet. Plant Physiol. 71, 350–355
Briskin, D.P., Thornley, R.W., Wyse, R.E. (1985a) Membrane transport in isolated vesicles from sugarbeet taproot. I. Isolation and characterization of energy-dependent H+-transporting vesicles. Plant Physiol. 78, 865–870
Briskin, D.P., Thornley, R.W., Wyse, R.E. (1985b) Membrane transport in isolated vesicles from sugarbeet taproot. II. Evidence for a sucrose/H+-antiport. Plant Physiol. 78, 871–875
Doll, S., Rodier, F., Willenbrink, J. (1979) Accumulation of sucrose in vacuoles isolated from red beet tissue. Planta 144, 407–411
Feige, B.H., Gimmler, H., Jeschke, W.D., Simonis, W. (1969) Eine Methode zur dünnschichtchromatographischen Auftrennung von 14C-und 32P-markierten Stoffwechselprodukten. J. Chromatogr. 41, 80–90
Getz, H.-P. (1987) Accumulation of sucrose in vacuoles released from isolated beet root protoplasts by both direct sucrose uptake and UDP-glucose-dependent group translocation. J. Plant Physiol. Biochem., in press
Hawker, J.S., Hatch, M.D. (1966) A specific sucrose phosphatase from plant tissues. Biochem. J. 99, 102–107
Joshi, J.G. (1982) Phosphoglucomutase from yeast. Methods Enzymol 89, 599–605
Leloir, L.F., Cardini, C.E. (1962) UDPG-fructose-6-phosphate transglucosylase. Methods Enzymol. 5, 170–171
Lin, W., Wagner, G.J., Siegelman, H.W., Hind, G. (1977) Membrane bound ATPase of intact vacuoles and tonoplasts isolated from mature plant tissue. Biochim. Biophys. Acta 465, 100–117
Maretzki, A., Thom, M. (1986) A group translocator for sucrose assimilation in tonoplast vesicles of sugarcane cells. Plant Physiol. 80, 34–37
Maretzki, A., Thom, M. (1987) UDP-glucose-dependent sucrose translocation in tonoplast vesicles from stalk tissue of sugar cane. Plant Physiol. 83, 235–237
Niemietz, C., Willenbrink, J. (1985) The function of tonoplast ATPase in intact vacuoles of red beet is governed by direct and indirect ion effects. Planta 166, 545–549
Poole, R.J., Briskin, D.P., Kratky, Z., Johnstone, R.M. (1984) Density gradient localization of plasma membrane and tonoplast from storage tissue of growing and dormant red beet. Plant Physiol. 74, 549–556
Salerno, G.L., Pontis, H.G. (1978) Studies of sucrose phosphate synthetase. The inhibitory action of sucrose. FEBS Lett. 86, 263–267
Sowokinos, J.R. (1976) Pyrophosphorylases in Solanum tuberosum. I. Changes in ADP-glucose and UDP-glucose pyrophosphorylase activities associated with starch biosynthesis during tuberization, maturation and storage of potatoes. Plant Physiol. 57, 63–68
Sze, H. (1985) H+-translocating ATPases: Advances using membrane vesicles. Annu. Rev. Plant Physiol. 36, 175–208
Thom, M., Komor, E. (1984) H+/sugar antiport as the mechanisms of sugar uptake by sugarcane vacuoles. FEBS Lett. 173, 1–4
Thom, M., Komor, E., Maretzki, A. (1982) Vacuoles from sugarcane suspension cultures. II. Characterization of sugar uptake. Plant Physiol. 69, 1320–1325
Thom, M., Maretzki, A. (1985) Group translocation as a mechanism for sucrose transfer into vacuoles from sugarcane cells. Proc. Natl. Acad. Sci. USA 82, 4697–4701
Thom, M., Leigh, R.A., Maretzki, A. (1986) Evidence for the involvement of an UDP-glucose-dependent group translocator in sucrose uptake into vacuoles of storage roots of red beet. Planta 167, 410–413
Willenbrink, J. (1987) Die pflanzliche Vakuole als Speicher. Naturwissenschaften 74, 22–29
Willenbrink, J., Doll, S., Getz, H.-P., Meyer, S. (1984) Zuckeraufnahme in isolierte Vakuolen and Protoplasten aus dem Speichergewebe von Beta-Rüben. Ber. Dtsch. Bot. Ges 97, 27–39
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Voß, M., Weidner, M. Uridine 5′-diphospho-D-glucose-dependent vectorial sucrose synthesis in tonoplast vesicles of the storage hypocotyl of red beet (Beta vulgaris L. ssp. conditiva). Planta 173, 96–103 (1988). https://doi.org/10.1007/BF00394493
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00394493