Summary
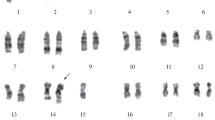
A mutant human lymphoblastoid cell line, Raji-TG, resistant to 10μg 6-thioguanine (TG)/ml was produced from wild-type cells after exposure to ethylmethane sulfonate. The Raji-TG cells showed their failure to incorporate 3H-hypoxanthine, only 2% as much hypoxanthine guanine phosphoribosyl transferase (HPRT) activity as wild-type cells, and no revertant in HAT selective medium containing hypoxanthine, aminopterin, and thymidine. Raji-TG cells, which were maintained routinely in regular medium lacking TG for as long as 2 years, still retained resistance to the drug and inability to grow in HAT medium. A fusion of Raji-TG cells and mouse cells resistant to 5-bromodeoxyuridine and lacking thymidine kinase formed hybrids, and the resulting hybrid colonies proliferated in HAT medium. These observations strongly supported the hypothesis that Raji-TG line cells might be originated from a mutational event with deficiency of HPRT. Both parental and the mutant have a modal chromosome number of 49 with a remarkably stable karyotype. Excess chromosome materials are found in chromosomes 1, 5, 7, 14, and 16. Chromosome 8 is completely missing, but is represented by two respective isochromosomes of the short and long arms of No. 8. Five different marker chromosomes could be distinguished, and most of their origin has been determined. Isolation of Raji-TG X mouse hybrid clones which contained one of each marker chromosome is of considerable value in mapping human genes on regions within particular chromosomes.
Similar content being viewed by others
References
Chu, E. H., Brimer, P., Jacobson, K. B., Merriam, E. V.: Mammalian cell genetics, I. Selection and characterization of mutation auxotrophic for L-glutamine or resistant to 8-azaguanine in Chinese hamster cells in vitro. Genetics 62, 359–377 (1969)
DeMars, R., Held, K. R.: The spontaneous azaguanine-resistant mutants of diploid human fibroblasts. Humangenetik 16, 87–110 (1972)
Gee, P. A., Ray, M., Mohandas, T., Douglas, G. R., Palser, H. R., Richardson, B. J., Harmerton, J. L.: Characteristics of an HPRT-deficient Chinese hamster cell line. Cytogenet. Cell Genet. 13, 437–447 (1974)
Gillin, F. D., Roufa, D. J., Beaudet, A. L., Caskey, C. T.: 8-azaguanine resistance in mammalian cells, I. Hypoxanthine-guanine phosphoribosyl transferase. Genetics 72, 239–252 (1972)
Harris, M.: Mutation rates in cells at different ploidy levels. J. Cell Physiol. 78, 177–184 (1971)
Hsie, A. W., Brimer, P. A., Mitchell, T. J., Gosslee, D. G.: The dose-response relationship for ethylmethansulfonate-induced mutations at the hypoxanthin-guanine phosphoribosyl transferase locus. Somatic Cell Genetics. 1, 247–261 (1975)
Littlefield, J. W.: The selection of hybrid mouse fibroblasts. cold Spring Harbor Symp. Quant. Biol. 29, 161–166 (1964)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
Manolov, G., Manolova, Y.: Marker band in one chromosome 14 from Burkitt lymphomas. Nature 237, 33–34 (1972)
Mezger-Freed, L.: Effect of ploidy and mutagens on bromodeoxyuridine resistance in haploid and diploid frog cells. Nature (New Biol.) 235, 245–246 (1972)
Morrow, J.: Genetic analysis of azaguanine resistance in an established mouse cell line. Genetics 65, 279–287 (1970)
New Haven Conference (1973): 1st Intern. Workshop on Hum. Gene Mapp. Birth Defects: Orig. Artic. Ser. X, 3. New York: The National Foundation 1974
Nyormoi, O., Sinclair, J. H., Klein, G.: Isolation and characterization of an adherent, 8-azaguanine resistant variant of the Burkitt lymphoma cell line. Raji. Expt. Cell Res. 82, 241–251 (1973)
Pasztor, L. M., Hu, F., Staukova, L., Bigley, R.: 8-azaguanine-resistant melanoma cells in vitro and in vivo. J. Natl. Cancer Inst 52, 1143–1150 (1974)
Pulvertaft, R. J. V.: Cytology of Burkitt's tumor (African Lymphoma). Lancet 1964I, 238–240
Ruddle, F. H., Creagan, R. P.: Parasexual approaches to the genetics of man. Ann. Rev. Genet. 9, 407–486 (1975)
Rotterdam Conference (1974): 2nd Intern. Workshop on Human Genetic Mapping Birth Defects: Orig. Artic. Ser. X. New York: The National Foundation 1975
Sato, K., Slesinski, R. S., Littlefield, J. W.: Chemical mutagenesis at the phosphoribosyl transferase locus in cultured human lymphoblasts. Proc. Natl. Acad. Sci. (USA) 69, 1244–1248 (1972)
Seabright, M.: A rapid banding technique for human chromosomes. Lancet 1971 II, 971–972
Steel, C. M.: Non-identity of apparently similar chromosome aberrations in human lymphoblastoid cell lines. Nature 233, 555–556 (1971)
Yoshida, M. C., Ikeuchi, T.: Enhancement of Sendai virus-mediated fusion and hybridization of lymphoid cells by addition of phytohemagglutinin. Proc. Jap. Acad. 51, 126–129 (1975)
Yoshida, M. C., Ikeuchi, T., Sasaki, M.: Differential staining of parental chromosomes in interspecific cell hybrids with a combined quinacrine and 33 258 Hoechst technique. Proc. Jap. Acad. 51, 184–187 (1975)
Yoshida, M. C., Matsuya, Y.: Confirmation of the human thymidine kinase locus, 17q21→17q ter, by means of a man-mouse somatic cell hybrid, D98/AH-2 X LMTK-Cl-1D. Hum. Genet. 31, 235–239 (1976)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshida, M.C., Kodama, Y. Karyological characterization of a human lymphoblastoid cell line resistant to 6-thioguanine. Hum Genet 35, 201–208 (1977). https://doi.org/10.1007/BF00393971
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00393971