Abstract
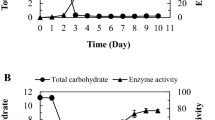
A dextran-hydrolysing enzyme from Lipomyces lipofer IGC 4042 was purified from the supernatant of cultures grown on a mineral medium with dextran, by ultrafiltration and gel filtration on Bio Gel A-0.5 m. This preparation gave only one band by disc gel electrophoresis. Glucose was the only product of dextran hydrolysis. Optimum pH and temperature for the activity of the enzyme were pH 4.5–5.0 and 45°C, respectively. The enzyme was most stable over a pH range of 4.5–6.0, and after 2 hours at 50°C maintained over 60% of its original activity. The molecular weight was 29,000 daltons and the isoelectric point was at pH 7. Km (45°C, pH 5) for dextran T-40 was 1.2×10−5 M. Glucose inhibited the enzyme competitively with a Ki (45°C, pH 5) of 0.5 mM.
Similar content being viewed by others
References
Andrews, P. 1964. Estimation of the molecular weights of proteins by Sephadex gel filtration.—Biochem. J. 91: 222–232.
Block, R. J., Durrum, E. L. and Zweig, G. 1958. A Manual of Paper Chromatography and Electrophoresis, 2nd ed.—Academic Press. New York.
Bonse, D. 1974. Polysaccharidabbau durch Hefen. Ph. D. Thesis.—Institut für Gärungsgewerbe und Biotechnologie, Berlin.
Covacevich, M. T. and Richards, G. N. 1978. Purification of intracellular dextranases and D-glucosidases from Pseudomonas UQM 733.—Carbohydr. Res. 64: 169–180.
Haglund, H. 1977. Isoelectric focusing in pH gradients.—L.K.B. Produkter AB, Sweden.
Hiromi, K., Kawai, M., Suetsugu, N., Nitta, Y., Hosotani, T., Nagao, A., Nakajima, T. and Ono, S. 1973. Kinetic studies on glucoamylase.—J. Biochem. 74: 935–943.
Lowry, O. H., Rosebrough, N. J., Farr, A. L. and Randall, R. J. 1951. Protein measurement with the Folin phenol reagent.—J. Biol. Chem. 193: 265–275.
Sawai, T. 1964. A possible capacity of Candida transglucosyl-amylase to hydrolyse dextran and nigeran.—Bull. Aichi Gakuchei Univ. Nat. Sciences 13: 25–40.
Sawai, T., Tohyama, T. and Natsume, T. 1978. Hydrolysis of fourteen native dextrans by Arthrobacter isomaltodextranase and correlation with dextran structure.—Carbohydr. Res. 66: 195–205.
Sawai, T., Yamaki, T. and Ohya, T. 1976. Purification and some properties of Arthrobacter globiformis exo-1,6-α-glucosidase.—Agr. Biol. Chem. 40: 1293–1299.
Sery, T. W. and Hehre, E. J. 1956. Degradation of dextrans by enzymes of intestinal bacteria. —Biochem. J. 71: 373–380.
Sidebotham, R. L. 1974. Dextrans.—Adv. Carbohydr. Chem. Biochem. 30: 371–433.
Sugiura, M., Ito, A. and Amaguchi, T. 1974. Studies on dextranase. II. New exo-dextranase from Brevibacterium fuscum var. dextranlyticum.—Biochim. Biophys. Acta 350: 61–70.
Torii, M., Sakakibara, K., Misaki, A. and Sawai, T. 1976. Degradation of alpha-linked D-glucoseoligosaccharides and dextrans by an isomalto-dextranase from Arthrobacter globiformis T6.—Biochem. Biophys. Res. Commun. 70: 459–464.
Tsuchiya, H. M., Jeanes, A., Bricker, H. M. and Wilham, C. A. 1952. Dextran-degrading enzymes from molds.—J. Bacteriol. 64: 513–519.
Van Uden, N. 1967. Transport-limited fermentation and growth of Saccharomyces cerevisiae and its competitive inhibition.—Arch. Mikrobiol. 58: 155–168.
Walker, G. J. and Pulkownik, A. 1973. Degradation of dextrans by an α-1,6-glucan glucohydrolase from Streptococcus mitis.—Carbohydr. Res. 29: 1–14.
Webb, E. and Spencer-Martins, I. Can. J. Microbiol. (submitted for publication).
Zevenhuizen, L. P. T. M. 1968. Cell-bound exodextranase of Bacillus species.—Carbohydr. Res. 6: 310–318.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ramos, A., Spencer-Martins, I. Extracellular glucose-producing exodextranase of the yeast Lipomyces lipofer . Antonie van Leeuwenhoek 49, 183–190 (1983). https://doi.org/10.1007/BF00393677
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00393677