Abstract
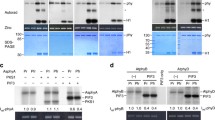
A protein-kinase activity which is co-purified with phytochrome from etiolated oat seedlings was investigated in some detail. Whereas phytochrome was always phosphorylated in solution (together with some contaminating protein bands), radioactive phosphate was not found in the phytochrome band after native gel electrophoresis and incubation of the entire gel with labeled ATP. Since protein kinases are usually autophosphorylated under these conditions, the result shows that the kinase activity does not reside in the phytochrome molecule itself. Radioactivity was exclusively detected in a band with the apparent molecular weight 450 kDa; sodium-dodecyl-sulfate gel electrophoresis revealed an apparent molecular weight of 60 kDa for the phosphorylated subunit. The N-terminal amino-acid sequence A L E S GA K LQ V P W was determined for this subunit which is a potential candidate for the protein kinase. The optimum conditions (pH, metal ion concentration) and kinetics of the phosphorylation reaction were determined. The presumed connection between proteinkinase activity and the signal chain leading from the far-red-absorbing form of phytochrome to physiological responses still awaits elucidation.
Similar content being viewed by others
Abbreviations
- Bistris:
-
2-[bis(2-hydroxyethyl)amino]-2-(hydroxymethyl)-1,3-propanediol
- kDa:
-
kilodalton
- Pfr:
-
far-red absorbing form of phytochrome
- Pr:
-
red-absorbing form of phytochrome
- PMBS:
-
p-chloromercuribenzenesulfonate
- SDS:
-
sodium dodecyl sulfate
- Tris:
-
2-amino-2-(hydroxymethyl)-1,3-propanediol
References
Bairoch, A., Claverie, J.-M. (1988) Sequence patterns in protein kinases. Nature 331, 22
Cordonnier, M.M., Greppin, H., Pratt, L.H. (1985) Monoclonal antibodies with differing affinities to the red- and far-red absorbing forms of phytochrome. Biochemistry 24, 3246–3523
Eckerskorn, Ch., Mewes, W., Goretzki, H., Lottspeich, F. (1988) A new siliconized glass fiber as support for proteinchemical analysis of electroblotted proteins. Eur. J. Biochem. 176, 509–519
Edelman, A.M., Blumenthal, D.K., Krebs, E.G. (1987) Protein serine/threonine kinases. Annu. Rev. Biochem. 56, 567–613
Eilfeld, P., Haupt, W. (1989) Phytochrome. In: Photoreceptor function and evolution. Holmes, M.G. ed., Academic Press, London, in press
Eilfeld, P.H., Widerer, G., Malinowski, H., Rüdiger, W., Eilfeld, P.G. (1988) Topography of the phytochrome molecule as determined from chemical modification of SH-groups. Z. Naturforsch. 43c, 63–73
Ernst, D., Oesterhelt, D. (1984) Purified phytochrome influences in vitro transcription in rye nuclei. EMBO J 3, 3075–3078
Ernst, D., Vojacek, R., Oesterhelt, D. (1987) Purification of phytochrome from rye by fast protein liquid chromatography. Photochem. Photobiol. 45, 859–862
Furuya, M. ed. (1987) Phytochrome and photoregulation in plants. Academic Press, Tokyo New York
Grimm, R., Eckerskorn, Ch., Lottspeich, F., Zenger, C., Rüdiger, W. (1988) Sequence analysis of proteolytic fragments of 124-kilodalton phytochrome from etiolated Avena sativa L.: Conclusions on the conformation of the native protein. Planta 174, 396–401
Grimm, R., Lottspeich, F., Rüdiger, W. (1987) Heterogeneity of the amino acid sequence of phytochrome from etiolated oat seedlings. FEBS Lett. 225, 215–217
Grimm, R., Lottspeich, F., Schneider, Hj.A.W., Rüdiger, W. (1986) Investigation of the peptide chain of 124 kDa phytochrome: localization of proteolytic fragments and epitopes for monoclonal antibodies. Z. Naturforsch. 41c, 993–1000
Grimm, R., Rüdiger, W. (1986) A simple and rapid method for isolation of 124 kDa oat phytochrome. Z. Naturforsch. 41c, 988–992
Hanks, S.K., Quinn, A.M., Hunter, T. (1988) The protein kinase family: Conserved features and deduced phylogeny of the catalytic domains. Science 241, 42–52
Hershey, H.P., Barker, R.F., Idler, K.B., Lissemore, J.L., Quail, P.H. (1985) Analysis of cloned cDNA and genomic sequences for phytochrome: Complete amino acid sequences for two gene products expressed in etiolated Avena. Nucleic Acids Res. 13, 8543–8559
Jones, A.M., Vierstra, R.D., Daniels, S.M., Quail, P. (1985) The role of separate molecular domains in the structure of phytochrome from etiolated Avena sativa L. Planta 164, 501–506
Kerscher, L. (1983) Subunit size, absorption spectra and dark reversion kinetics of rye phytochrome purified in the far-red absorbing form. Plant Sci. Lett. 32, 133–138
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685
Lagarias, J.G., Mercurio, F.M. (1985) Structure function studies on phytochrome. Identification of light-induced conformational changes in 124 kDa Avena phytochrome in vitro. J. Biol. Chem. 260, 2415–2423
Lagarias, J.C., Wong, Y.-S., Berkelman, T.R., Kidd, D.G., McMichael, R.W., Jr. (1987) Structure-function studies on Avena phytochrome. In: Phytochrome and photoregulation in plants, pp. 51–61, Furuya, M., ed. Academic Press, Tokyo New York
Litts, J.C., Kelly, J.M., Lagarias, J.C. (1983) Structure-function studies on phytochrome. Preliminary characterization of highly purified phytochrome from Avena sativa enriched in the 124-kilodaltion species. J. Biol. Chem 258, 11025–11031
Shanklin, J., Jabben, M., Vierstra, R.D. (1987) Red light-induced formation of ubiquitin-phytochrome conjugates: Identification of possible intermediates of phytochrome degradation. Proc. Natl. Acad. Sci. USA 84, 359–363
Soll, J. (1985) Phosphoproteins and protein kinase activity in isolated envelopes of pea chloroplasts. Planta 166, 394–400
Soll, J. (1988) Purification and characterization of a chloroplast outer-envelope-bound, ATP-dependent protein kinase. Plant Physiol. 87, 898–903
Speth, V., Otto, V., Schäfer, E. (1987) Intracellular localisation of phytochrome and ubiquitin in red-light-irradiated oat coleoptiles by electron microscopy. Planta 171, 332–338
Vierstra, R.D., Quail, P.H., Hahn, T.-R., Song, P.-S. (1987) Comparison of the protein conformations between different forms (Pr and Pfr) of native (124 kDa) and degraded (118/114 kDa) phytochrome from Avena sativa. Photochem. Photobiol. 45, 429–432
Wong, Y.-S., Cheng, H.-C., Walsh, D.A., Lagarias, J.C. (1986) Phosphorylation of Avena phytochrome in vitro as probe of light-induced conformational changes. J. Biol. Chem. 261, 12089–12097
Author information
Authors and Affiliations
Additional information
Dedicated to Professor A. Trebst on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Grimm, R., Gast, D. & Rüdiger, W. Characterization of a protein-kinase activity associated with phytochrome from etiolated oat (Avena sativa L.) seedlings. Planta 178, 199–206 (1989). https://doi.org/10.1007/BF00393195
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00393195