Abstract
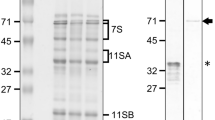
The synthesis and deposition of the endosperm storage proteins of rye, usually termed secalins, has been studied. The rate of accumulation of secalin in developing rye grain was at a maximum between 3 and 5 weeks after anthesis. Some changes in the proportions of the four major groups of secalin polypeptides were observed during maturation, notably an increase in γ-secalins of Mr 75k and a decrease in ω-secalins. In-vitro translation of mRNA fractions prepared from 4-week-old endosperms showed that secalin polypeptides were synthesised on membrane-bound polysomes. The secalin products were identified by their mobilities on SDS-PAGE and their relative incorporation of radioactive lysine, glycine, proline, leucine and methionine. Protein bodies prepared by sucrose density ultracentrifugation contained reduced amounts of γ-secalins of Mr 40k and ω-secalins compared with the total secalin fraction, but these components were present in the expected amounts when 1.0 M NaCl was added to the buffers. Treatment of the protein bodies with proteinase-k resulted in the digestion of their contents regardless of the presence of NaCl, indicating that the surrounding membrane was incomplete. It was concluded that the NaCl reduced the loss of secalins from the protein bodies by decreasing secalin solubility rather than by affecting the integrity of the protein body membrane. The results reported for the synthesis and deposition of secalins are consistent with the results of previous studies on the prolamins of wheat and barley.
Similar content being viewed by others
Abbreviations
- HMW:
-
high molecular weight
- SDS-PAGE:
-
sodium-dodecylsulphate polyacrylamide-gel electrophoresis
References
Bietz, J.A., Huebner, F.R., Sandersen, J.E., Wall, J.S. (1977) Wheat gliadin homology revealed through N-terminal amino acid sequence analysis. Cereal Chem. 54, 1070–1083
Brandt, A. (1976) Endosperm protein formation during kernal development of wild type and a high lysine barley mutant. Cereal Chem. 53, 890–901
Brandt, A., Ingversen, J. (1976) In vitro synthesis of barley endosperm proteins on wild type and mutant templates. Carlsberg Res. Commun. 41, 311–320
Cameron-Mills, V. (1980) The structure and composition of protein bodies purified from barley endosperm by silica sol density gradients. Carlsberg Res. Commun. 45, 557–576
Chamberlain, J.P. (1979) Fluorographic detection of radioactivity in polyacrylamide gels with the water-soluble fluor, sodium salicylate. Anal. Biochem. 98, 132–135
Charbonnier, L., Terce-Lafogue, T., Mosse, J. (1981) Rye prolamins: Extractability, separation and characterization. J. Agric. Food Chem. 29, 968–973
Donovan, G.R., Lews, J.W., Longhurst, T.J. (1982) Cell-free synthesis of wheat prolamins. Aust. J. Plant Physiol. 9, 59–68
Field, J.M., Shewry, P.R., Miflin, B.J., March, J.F. (1982) The purification and characterization of homologous high molecular weight storage proteins from grain of wheat, rye and barley. Theor. Appl. Genet. 62, 329–336
Flint, D., Ayers, G.S., Ries, S.K. (1975) Synthesis of endosperm proteins in wheat seed during maturation. Plant Physiol. 56, 381–384
Forde, B.G., Kreis, M., Bahramian, M.B., Matthews, J.A., Miflin, B.J., Thompson, R.D., Bartels, D., Flavell, R.B. (1981) Molecular cloning and analysis of cDNA sequences derived from poly(A)+RNA from barley endosperm: identification of B hordein related clones. Nucl. Acids Res. 9, 6689–6707
Forde, J., Miflin, B.J. (1983) Isolation and identification of mRNA for the high-molecular-weight storage proteins of wheat endosperm. Planta 157, 567–576
Fox, J.E., Pratt, H.M., Shewry, P.R., Miflin, B.J. (1977) The in vitro synthesis of hordeins with polysomes from normal and high-lysine varieties of barley. In: Nicleic acids and protein synthesis in plants, pp. 501–509. Centre National de la Recherche Scientifique, Paris
Friedman, M., Krull, L.H., Cavins, J.F. (1970) The chromatographic determination of cystine and cysteine residues as S-β-(4-pyridylethyl) cysteine. J. Biol. Chem. 245, 3868–3871
Greene, F.C. (1981) In vitro synthesis of wheat (Triticum aestivum L.) storage proteins. Plant Physiol. 68, 778–783
Kasarda, D.D., Autran, J-C., Lew, E. J-L., Nimmo, C.C., Shewry, P.R. (1983) N-terminal amino acid sequences of ω-gliadins and ω-secalins: implications for the evolution of prolamin genes. Biochim. Biophys. Acta 747, 138–150
Kasarda, D.D., Bernardin, J.E., Nimmo, C.C. (1976) Wheat proteins. In: Advances in cereal science and technology, vol. 1, pp. 158–236, Pomeranz Y., ed. Am. Assoc. Cereal Chem., St. Paul, Minn., USA
Laemmli, V.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227, 681–685
Matthews, J.A., Miflin, B.J. (1980) In vitro synthesis of barley storage proteins. Planta 149, 262–268
Mecham, D.K., Fullington, J.G., Greene, F.C. (1981) Gliadin proteins in the developing wheat seed. J. Sci. Food Agric. 32, 773–780
Miflin, B.J., Burgess, S.R. (1982) Protein bodies from developing seeds of barley, maize, wheat and peas — effects of protease treatment. J. Exp. Bot. 33, 251–260
Miflin, B.J., Burgess, S.R., Shewry, P.R. (1981) The development of protein bodies in the storage tissues of seeds. J. Exp. Bot. 32, 199–219
Miflin, B.J., Field, J.M., Shewry, P.R. (1983) Cereal storage proteins and their effects on technological properties. In: Seed proteins, pp. 255–319, Daussant, J., Mosse, J., Vaughan, J., eds. Academic Press, London
Quail, P.H. (1979) Plant cell fractionation. Annu. Rev. Plant Physiol. 30, 425–484
Rahman, S., Shewry, P.R., Miflin, B.J. (1982) Differential protein accumulation during barley grain development. J. Exp. Bot. 33, 717–728
Roberts, B.E., Patterson, B.M. (1973) Efficient translation of tobacco mosaic virus RNA and rabbit globin 9S RNA in cell-free system from commercial wheat-germ. Proc. Natl. Acad. Sci. USA 70, 2330–2334
Schramm, M., Eisenkrat, F.B., Barkai, E. (1967) Cold-induced leakage of amylase from the zymogen granule and sealing of its membranes by specific lipids. Biochim. Biophys. Acta 135, 44–52
Shewry, P.R., Pratt, H.M., Leggatt, M.M., Miflin, B.J. (1979) Protein metabolism in developing endosperms of high-lysine and normal barley. Cereal Chem. 56, 110–117
Shewry, P.R., Autran, J.C., Nimmo, C.C., Lew, E.J.-L., Kasarda, D.D. (1980) N-terminal amino acid sequence homology of storage protein components from barley and a diploid wheat. Nature (London) 286, 520–522
Shewry, P.R., Field, J.M., Lew, E.J-L., Kasarda, D.D. (1982) The purification and characterization of two groups of storage proteins (secalins) from rye (Secale cerele L.). J. Exp. Bot. 133, 261–268
Shewry, P.R., Miflin, B.J. (1983) Characterization of and synthesis of barley seed proteins. In: Seed proteins. Biochemistry, Genetics, nutritive value, pp. 143–205, Gottschalk, W., Muller, H., eds. Martinus Nijhoff, The Netherlands
Shewry, P.R., Kasarda, D.D., Miflin, B.J. (1983a) Structural and evolutionary relationships of the prolamin storage proteins of barley, rey and wheat. Phil. Trans. R. Soc. London Ser. B (in press)
Shewry, P.R., Parmar, S., Miflin, B.J. (1983b) The extraction, separation and polymorphism of the prolamin storage proteins (secalins) of rye. Cereal Chem. 60, 1–6
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shewry, P.R., Kreis, M., Burgess, S.R. et al. The synthesis and deposition of the prolamin storage proteins (secalins) of rye. Planta 159, 439–445 (1983). https://doi.org/10.1007/BF00392080
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392080