Abstract
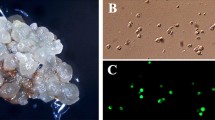
Rice (Oryza sativa L.) plants of the indica cultivar IR54 were regenerated from protoplasts. Conditions were developed for isolating and purifying protoplasts from suspension cultures with protoplast yields ranging from 1·106 to 15·106 viable protoplasts/1 g fresh weight. Protoplast viability after purification was generally over 90%. Protoplasts were cultured in a slightly modified Kao medium in a Petri plate by placing them onto a Millipore filter positioned on top of a feeder (nurse) culture containing cells from a suspension culture of the japonica rice, Calrose 76. Plating efficiencies of protoplasts ranged from 0.5 to 3.0%; it was zero in the absence of the nurse culture. Protoplast preparations usually contained no contaminating cells, and when present, the number of cells never exceeded 0.1% of the protoplasts. After three weeks the Millipore filter with callus colonies were transferred off feeder cells and onto a Linsmaier and Skoog-type medium for an additional three weeks. Selected callus colonies that had embryo-like structures were then transferred to regeneration medium containing cytokinins, and regeneration frequencies up to 80% were obtained. Small shoots emerged and were transferred to jars for root development prior to transferring to pots of soil and growing the plants to maturity in growth chambers. Of the cytokinins evaluated, N6-benzylaminopurine was the most effective in promoting shoot formation; however, kinetin was also somewhat effective. Regeneration medium could be either an N6 or Murashige and Skoog basal medium. Of 76 plants grown to maturity, 62 were fertile, and the plant heights averaged about three-fourths the height of seed-grown plants.
Two other suspension cultures of IR54, one developed from the protoplast callus of the initial IR54 line, and the other developed from callus produced by mature seeds, have yielded protoplasts capable of regenerating plants when using cells of the Calrose 76 suspension as a nurse culture. In addition, protoplasts obtained from three-week-old primary callus of immature embryos of IR54 were capable of regenerating plants when using the same culture conditions.
Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- pcy:
-
packed cell volume
- BAP:
-
N6-benzylaminopurine
- FDA:
-
fluorescein diacetate
- FW:
-
fresh weight
- IAA:
-
indole-3-acetic acid
- AA:
-
Muller and Grafe (1978)
- CPW:
-
Frearson et al. (1973)
- Kao* :
-
Kao (1977)
- LS:
-
Linsmaier and Skoog (1965)
- MS:
-
Murashige and Skoog (1962)
- N6 :
-
Chu et al. (1975)
- PCM:
-
Ludwig et al. (1985)
References
Abdullah, R., Cocking, E.C., Thompson, J.A. (1986) Efficient plant regeneration from rice protoplasts through somatic embryogenesis. Bio/Technology 4, 1087–1090
Chu, C.C., Wang, C.C., Sun, C.S., Hus, C., Yin, K.C., Chu, C.Y. (1975) Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Sci. Sin. 18, 659–668
Fujimura, T., Sakurai, M., Akagi, H., Negishi, T., Hirose, A. (1985) Regeneration of rice plants from protoplasts. Plant Tissue Cult. Lett. 2, 74–75
Frearson, E.M., Power, J.B., Cocking, E.C. (1973) The isolation, culture and regeneration of Petunia leaf protoplasts. Dev. Biol. 33, 130–137
Galbraith, D.W. (1981) Microfluorimetric quantitation of cellulose biosynthesis by plant protoplasts using calcofluor white. Physiol. Plant. 53, 111–116
Imbrie-Milligan, C.W., Hodges, T.K. (1986) Microcallus formation from maize protoplasts prepared from embryogenic callus. Planta 168, 395–401
Kamo, K.K., Chang, K.L., Lynn, M.E., Hodges, T.K. (1987) Embryogenic callus formation from maize protoplasts. Planta 172, 245–251
Kao, K.N. (1977) Chromosomal behavior in somatic hybrids of soybean — Nicotiana glauca. Mol. Gen. Genet. 150, 225–230
Koetje, D.S., Grimes, H.D., Wang, Y.C., Hodges, T.K. (1989) Regeneration of indica rice (Oryza sativa L.) from primary callus derived from immature embryos. J. Plant Physiol., in press
Kyozuka, J., Hayashi, Y., Shimamoto, K. (1987) High frequency plant regeneration from rice protoplasts by novel nurse culture methods. Mol. Gen. Genet. 206, 408–413
Linsmaier, E.M., Skoog, F. (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol. Plant. 18, 100–127
Ludwig, S.R., Somers, D.A., Petersen, W.L., Pohlman, R.F., Zarowitz, M.A., Gengenbach, B.G., Messing, J. (1985) High frequency callus formation from maize protoplasts. Theor. Appl. Genet. 71, 344–350
Muller, A.J., Grafe, R. (1978) Isolation and characterization of cell lines of Nicotiana tabacum lacking nitrate reductase. Mol. Gen. Genet. 161, 67–76
Murashige, T., Skoog, F. (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15, 473–497
Toriyama, K., Hinata, K., Sasaki, T. (1986) Haploid and diploid plant regeneration from protoplasts of anther callus in rice. Theor. Appl. Genet. 73, 16–19
Toriyama, K., Arimoto, Y., Uchimiya, H., Hinata, K. (1988) Transgenic rice plants after direct gene transfer into protoplasts. Bio/Technology 6, 1072–1074
Widholm, J.M. (1972) The use of fluorescein diacetate and phenosafranine for determining viability of cultured plant cells. Stain Technol. 47, 189–194
Yamada, Y., Yang, Z.Q., Tang, D.T. (1986) Plant regeneration from protoplast-derived callus of rice (Oryza sativa L). Plant Cell Rep. 4, 85–88
Zhang, H.M., Yang, H., Rech, E.L., Golds, T.J., Davis, A.S., Mulligan, B.J., Cocking, E.C., Davey, M.R. (1988) Transgenic rice plants produced by electroporation-mediated plasmid uptake into protoplasts. Plant Cell Rep. 7, 379–384
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, L., Schroll, R.E., Grimes, H.D. et al. Plant regeneration from indica rice (Oryza sativa L.) protoplasts. Planta 178, 325–333 (1989). https://doi.org/10.1007/BF00391860
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00391860