Summary
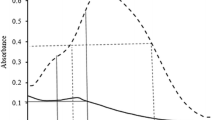
The enzymes α-amylase (α-1, 4-glucan 4-glucanohydrolase, 3.2.1.1), β-amylase (α-1,4-glucan maltohydrolase, 3.2.1.2) and phosphorylase (α-1,4-glucan: orthophosphate glucosyltransferase, 2.4.1.1) were assayed in whole grains of barley throughout the maturation period. α-amylase and phosphorylase had peaks of activity between 25 and 30 days after anthesis. On the other hand the activity of β-amylase in both the available and latent forms reached a maximum value at 35 days after anthesis which did not decrease thereafter. β-amylase activity was also assayed throughout development in the endosperm, aleurone, testa pericarp and embryo. Latent β-amylase reached a constant maximum value in endosperm at 35 days but available β-amylase reached a peak of activity at 25 days and then declined to zero at 45 days. Only latent β-amylase was associated with the aleurone layer and activity rose to a maximum value at 35 days. The testa pericarp had mainly latent β-amylase whose activity fell from an early maximum at 21 days to zero at 35 days. No hydrolytic activity was associated with the embryo. The phosphorylase activity was low and mainly associated with the endosperm fraction.
Similar content being viewed by others
References
Baxter, E. D.: Biochemistry of the developing cereal grain. Ph. D. Thesis, University of Edinburgh (1972)
Baxter, E. D., Duffus, C. M.: Starch synthetase in developing barley amyloplasts. Phytochemistry 10, 2641–2644 (1971).
Bernfeld, P.: Enzymes of starch degradation and synthesis. Advanc. Enzymol. 12, 379–428 (1951).
Bilderback, D. E.: Amylases in developing barley seeds. Plant Physiol. 48, 331–334 (1971).
Blom, J., Bak, A., Braae, B.: Untersuchungen über den enzymatischen Abbau der Stärke. Hoppe-Seylers Z. physiol. Chem. 241, 273–287 (1936).
Briggs, D. E.: A modification of the Sandstedt, Kneen and Blish assay of α-amylase. J. Inst. Brewing 67, 427–431 (1961).
Briggs, D. E.: Modified assay for α-amylase in germinating barley. J. Inst. Brewing 73, 361–370 (1967).
Cori, G. T., Colowick, S. P., Cori, C. F.: The formation of glucose-1-phosphoric acid in extracts of mammalian tissues and of yeast. J. biol. Chem. 123, 375–389 (1938).
Duffus, C. M.: α-amylase activity in the developing barley grain and its dependence on gibberellic acid. Phytochemistry 8, 1205–1209 (1969a).
Duffus, J. H.: Simplified method for calculating the results obtained with Briggs' α-amylase assay. J. Inst. Brewing 75, 252–253 (1969b).
Ford, J. S., Guthrie, J. M.: Contributions to the biochemistry of barley. J. Inst. Brewing 14, 61–87 (1908).
Gibson, R. A., Paleg, L. G.: Lysosomal nature of hormonally induced enzymes in wheat aleurone cells. Biochem. J. 128, 367–375 (1972).
LaBerge, D. E., Macgregor, A. W., Meredith, W. O. S.: Changes in α and β-amylase activities during the maturation of different barley cultivars. Canad. J. Plant Sci. 51, 469–477 (1971).
Merritt, N. R., Walker, J. T.: Development of starch and other components in normal and high amylase barley. J. Inst. Brewing 75, 156–164 (1969).
Oikawa, A.: The role of calcium in Taka-amylase A. II. The exchange reaction of calcium. J. Biochem. 46, 463–473 (1959).
Thompson, W. P., Johnston, D.: The cause of incompatability between barley and rye. Canad. J. Res. 23, Sec. C, 1–15 (1945).
Varner, J. E.: Gibberellic acid controlled synthesis of α-amylase in barley endosperm. Plant Physiol. 39, 413–415 (1964).
Weichherz, J., Asmus, R.: Untersuchungen über die enzymatischen Vorgänge bei der keimenden Gerste. Biochem. 237, 20–72 (1931).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Duffus, C., Rosie, R. Starch hydrolysing enzymes in the developing barley grain. Planta 109, 153–160 (1973). https://doi.org/10.1007/BF00386123
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00386123