Summary
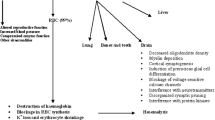
The effects of lead on red blood cell (RBC) membrane proteins were studied in two groups of workers with different lead exposure levels: Group 1 (6 subjects employed in a battery plant) with a mean blood lead of 40.1 (SD = 3.7) μg/100 ml; Group II(5 workers employed in different industries) with a mean blood lead of 60.6 (SD = 8.0) μg/100 ml, compared with a control group with mean blood lead of 15.6 (SD = 9.3) μg/100 ml. The analysis of RBC membrane polypeptides was carried out by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and by using a densitometer for percentage measurement of the bands corresponding to protein fractions. The results show a very significant decrease in Band 3 (anion channel) and 4.1 in more exposed workers (Group II) only. The effects of lead on RBC membrane proteins seem to be evident at blood-lead levels higher (> 50 μg/100 ml) than those previously reported in literature. These results confirm the effects of lead on membrane proteins, even if the exact mechanism, particularly the influence of proteolysis and the meaning of the interference, still needs to be investigated thoroughly.
Similar content being viewed by others
References
Apostoli P, Romeo L, Aprili F, Ferrari S (1985) Interference of lead on membrane lipids. Med Lav 76:485–490
Beutler E, West C, Blume KG (1976) The removal of leukocytes and platelets from whole blood. J Lab Clin Med 88:323–333
Cohen CM (1983) The molecular organization of the red cell membrane skeleton. Semin Hematol 20:141–158
Fairbanks G, Steck TL, Wallach DFH (1971) Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry 10:2606–2617
Fukumoto K, Karai I, Horiguchi S (1983) Effect of lead on erythrocyte membranes. Br J Ind Med 40:220–223
Goldberg A, Boches FS (1982) Oxidized proteins in erythrocytes are rapidly degraded by the adenosine triphosphatedependent proteolytic system. Science 215:1107–1109
Hasan J, Vihko V, Hernberg S (1967) Deficient red cell membrane Na/K ATPase in lead poisoning. Arch Environ Health 14:313–318
Hernberg S, Vihko V, Hasan J (1967) Red cell membrane ATPase in workers exposed to inorganic lead. Arch Environ Health 14:319–324
Karai I, Fukumoto K, Horiguchi S (1981) Studies on osmotic fragility of red blood cells determined with a Coil Planet Centrifuge for workers occupationally exposed to lead. Int Arch Occup Environ Health 48:273–281
Karai I, Fukumoto K, Horiguchi S (1982) Alterations of lipids of the erythrocyte membranes in workers exposed to lead. Int Arch Occup Environ Health 50:11–16
Karai I, Fukumoto K, Horiguchi S (1982) An increase in Na/K ATPase activity of erythrocyte membranes in workers employed in a lead refining factory. Br J Ind Med 39:290–294
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage. Nature 227:680–685
Mohandas N, Chasis JA, Shohet SB (1983) The influence of membrane skeleton on red cell deformability, membrane material properties, and shape. Semin Hematol 20:225–242
Ong CN, Lee WR (1980) Distribution of lead-203 in human peripheral blood in vitro. Br J Ind Med 37:78–84
Pontremoli S, Melloni E, Salamino F, Sparatore B, Benatti U, Morelli A, De Flora A (1980) Identification of proteolytic activities in the cytosolic compartement of mature human erythrocytes. Eur J Biochem 110:421–430
Secchi GC, Alessio L (1969) Studies on the mechanism of the inhibition by lead of erythrocyte Na+/K+-ATPase. Med Lav 60:670–673
Secchi GC, Rezzonico A, Alessio L (1968) Changes in Na+-K+-ATPase activity of erythrocytic membranes in different phases of lead poisoning. Med Lav 22:191–196
Sheetz MP, Singer SJ (1977) On the mechanism of ATP-induced shape changes in human erythrocyte membrane. J Cell Biol 73:638–646
Valentino M, Fiorini RM, Curatola G, Governa M (1982) Changes of membrane fluidity in erythrocytes of lead exposed workers. Int Arch Occup Environ Health 51:105–112
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Apostoli, P., Romeo, L., De Matteis, M.C. et al. Effects of lead on red blood cell membrane proteins. Int. Arch Occup Environ Heath 61, 71–75 (1988). https://doi.org/10.1007/BF00381610
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00381610