Summary
-
1.
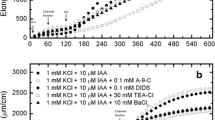
The acid-growth theory predicts that the rates of auxin-induced cell elongation and H+-excretion should be closely correlated as long as the experimental conditions remain fairly constant. To test this, Avena coleoptiles have been induced to elongate at different rates by varying the concentration of auxin, the age of the tissue, or by addition of metabolic inhibitors. As predicted, in each case there was a close correlation between the rates of H+-excretion and growth.
-
2.
The rate and direction of movement of H+ between the coleoptile and the external medium is regulated by the external pH. Coleoptiles take up H+ passively from acidic solutions and excrete H+ into basic solutions. In the absence of auxin, uptake and excretion are at equilibrium when the solution pH is near 5.7, a pH too high to allow rapid cell wall loosening. Auxin stimulates the excretion, but as the external pH drops the excretion is inhibited and a new equilibrium is established near 5.0. This allows amlows maximum wall loosening without causing toxic side-effects.
-
3.
H+-excretion is also affected by water stress. Increasing water stress induced by mannitol decreases H+-excretion in auxin-treated tissues but stimulates H+-excretion in the absence of auxin. At incipient plasmolysis H+-excretion is insensitive to auxin, suggesting that even if H+-excretion is mediated by an auxin-activated ATPase it may be impossible to demonstrate an effect of auxin on this enzyme in vitro.
-
4.
Three types of H+-excretion have been recognized in coleoptiles; auxin-induced, stress-induced, and basal level. All appear to require ATP, but only basal level H+-excretion does not also require continual protein synthesis.
Similar content being viewed by others
Abbreviations
- CCCP:
-
carbonyl cyanide m-chlorophenyl-hydrazone
- CHI:
-
cycloheximide
- IAA:
-
indoleacetic acid
References
Andrea, K.: Die auxininduzierte pH-Erniedrigung im extrazellulären Raum. 66 pp. Univ. Münster: Masters Thesis 1974
Cleland, R.: Cell wall extension. Ann. Rev. Plant Physiol. 22, 197–222 (1971a)
Cleland, R.: Instability of the growth-limiting proteins of the Avena coleoptile and their pool size in relation to auxin. Planta (Berl.) 99, 1–11 (1971b)
Cleland, R.: Dosage response curve for auxin-induced cell elongation: a reevaluation. Planta (Berl.) 104, 1–9 (1972)
Cleland, R.: Auxin-induced hydrogen ion excretion from Avena coleoptiles. Proc. nat. Acad. Sci. (Wash.) 70, 3092–3093 (1973)
Hager, A., Menzel, H., Krauss, A.: Versuche und Hypothese zur Primärwirkung des Auxins beim Streckungswachstum. Planta (Berl.) 100, 47–75 (1971)
Hamilton, T. H., Moore, R. J., Rumsey, A. F., Means, A. R., Schrank, A. R.: Stimulation of synthesis of ribonucleic acid in sub-apical sections of Avena coleoptile by IAA. Nature (Lond.) 208, 1180–1183 (1965)
Higinbotham, N.: Electropotentials of plant cells. Ann. Rev. Plant Physiol. 24, 25–46 (1973)
Hodges, T. K., Leonard, R. T., Bracker, C. E., Keenan, T. W.: Purification of an ion-stimulated ATPase from plant roots: association with plasma membrane. Proc. nat. Acad. Sci. (Wash.) 69, 3307–3311 (1972)
Ilan, I.: On auxin-induced pH drop and on the improbability of its involvement in the primary mechanism of auxin-induced growth promotion. Physiol. Plant. 28, 146–148 (1973)
Kasamo, K., Yamaki, T.: Effect of auxin in Mg++-activated and-inhibited ATPase from mung bean hypocotyls. Plant Cell Physiol. 15, 957–964 (1974)
Kitasato, H.: The influence of H+ on the membrane potential and ion fluxes in Nitella. J. Gen. Physiol. 52, 60–87 (1968)
Lai, Y. F., Thompson, J. E.: The preparation and properties of an isolated plant membrane fraction enriched in (Na+−K+)-stimulated ATPase. Biochim. biophys. Acta (Amst.) 233, 84–90 (1971)
MacDonald, I. R., Ellis, R. J.: Does cycloheximide inhibit protein synthesis specifically in plant tissues? Nature New Biol. 222, 791–792 (1969)
McMahon, D.: Cycloheximide is not a specific inhibitor of protein synthesis in vivo. Plant Physiol. 55, 815–821 (1975)
Marrè, E., Lado, P., Rasi Caldogno, F., Colombo, R.: Correlation between cell enlargement in pea internode segments and decrease in the pH of the medium of incubation. I. Effects of fusicoccin, natural and synthetic auxins and mannitol. Plant Sci. Letters 1, 179–184 (1973)
Marrè, E., Colombo, R., Lado, P., Rasi Caldogno, F.: Correlation between proton extrusion and stimulation of cell enlargement. Effects of fusicoccin and of cytokinins on leaf fragments and isolated cotyledons. Plant Sci. Letters 2, 139–150 (1974)
Penny, P., Dunlop, J., Perley, J. E., Penny, D.: pH and auxin-induced growth: a causal relationship? Plant Sci. Letters 4, 35–40 (1975)
Pitman, M. G.: Active H+ efflux from cells of low-salt barley roots during salt accumulation. Plant Physiol. 45, 787–790 (1970)
Raven, J. A., Smith, F. A.: Significance of hydrogen ion transport in plant cells. Can. J. Bot. 52, 1035–1048 (1974)
Rayle, D. L.: Auxin-induced hydrogen-ion secretion in Avena coleoptiles and its implications. Planta (Berl.) 114, 68–73 (1973)
Rayle, D. L., Cleland, R.: Enhancement of wall loosening and elongation by acid solutions. Plant Physiol. 46, 250–253 (1970)
Rayle, D. L., Cleland, R.: The in vitro acid-growth response: relation to in vivo growth responses and auxin action. Planta (Berl.) 104, 282–296 (1972)
Saito, K., Senda, M.: The electrogenic ion pump revealed by the external pH effect on the membrane potential of Nitella. Influences of external ions and electric current on the pH effect. Plant Cell Physiol. 15, 1007–1016 (1974)
Slayman, C. L.: Movement of ions and electrogenesis in micro-organisms. Amer. Zool. 10, 377–392 (1970)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cleland, R.E. Auxin-induced hydrogen ion excretion: correlation with growth, and control by external pH and water stress. Planta 127, 233–242 (1975). https://doi.org/10.1007/BF00380720
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00380720