Summary
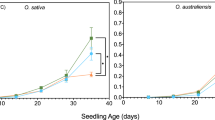
Kinetic properties of NAD malate dehydrogenase (MDH) and glutamate oxaloacetate transaminase (GOT) were analyzed in two genotypes of Arabidopsis thaliana collected in two sites of contrasting climates. Plants from each genotype were acclimated under controlled conditions at four different thermoperiods: 5–10° C, 7–15° C, 15–25° C and 25–28° C. Apparent energy of activation for MDH of the cold adapted genotype were significantly lower at low temperatures of acclimation, while for GOT, significant differences were found but no clear patterns emerge from the data. No differences of significance between the two genotypes were observed for apparent K m and K cat of both enzymes. For MDH, apparent K ms for oxaloacetic acid increased as a positive function of assay temperature but for GOT, K ms for α-oxoglutaric acid did not vary significantly over the 10–35° C assay temperature range. K cat values for both enzymes increased about 2 fold for every 10° C raise in assay temperature. Concentrations of both enzymes significantly increased in plants of both genotypes acclimated to the coldest thermoperiod. The concentration of GOT was signficantly higher in plants of the cold adapted genotype acclimated to 5–10° C and 7–15° C. Results suggest that MDH and GOT from the cold adapted genotype are more efficient in the modulation of catalysis at low temperatures, while the opposite is found for plants of the warm-adapted genotype through enhanced thermostability of the mitochondrial fraction of MDH.
Similar content being viewed by others
References
Arakawa Y, Katoh A, Sasaki R, Youshida K (1980) Purification and properties of glutamic oxaloacetate transaminase from pig liver. Nihon Univ J Med 22:163–180
Banaszak LJ, Bradshaw RA (1975) Malate dehydrogenases. In The Enzymes Boyer PD (ed) Vol XI. Academic Press, New York, pp 369–396
Bergmeyer H-V, Bernt E (1974) Glutamic oxaloacetate Transaminase. In Methods of enzymatic analysis (Bergmeyer H-V) (ed) Academic Press, New York, pp 727–751
Braunstein AE (1973) Amino group transfer. In “The Enzymes” Boyer PD (ed) Vol IX. Academic Press, New York, pp 379–481
Brouillet L, Simon J-P (1980) Adaptation and acclimation of higher plants at the enzyme level: Thermal properties of NAD malate dehydrogenase of two species of Aster (Asteraceae) and their hybrid adapted to contrasting habitats. Can J Bot 58:1474–1481
Chabot BF (1979) Metabolic and enzymatic adaptations to low temperature. In: Underwood LF, Tieszen LL, Callahan AB, Folk GE (eds) Comparative mechanisms of cold adaptation. Academic Press, NY, pp 283–301
Davidson D, Simon J-P (1981) Thermal adaptation and acclimation of ecotypic populations of Spirodela polyrhiza (L.) Schleid. (Lemnaceae): Thermostability and apparent activation energy of NAD malate dehydrogenase. Can J Bot 59:1061–1068
Davidson D, Simon J-P (1983) Thermal adaptation and acclimation of ecotypic populations of Spirodela polyrhiza (L.) Schleid.: temperature dependency of K m of NAD malate dehydrogenase. J Thermal Biol 8:289–296
Gupta DN, Roy MK, Singh JN, Singhal GS, Mehta SL (1980) Changes in proteins and enzymes during grain development in pearl millet. Biochem Physiol Planz 175:15–22
Hazel JR, Prosser CL (1974) Molecular mechanisms of temperature compensation in poikilotherms. Physiol Rev 54:620–677
Hochachka PW, Somero GN (1973) Strategies of biochemical adaptation. W.B. Saunders Co., Philadelphia
Johnson GB (1979) Enzyme polymorphism: Genetic variation and the physiological phenotype in Topics in plant population Biology Solbrig OT, Jain SH, Johnson GB, Raven PH (eds) Columbia Univ Press, New York, 62–82
Jones JC, Hancock JF, Liu EH (1979) Biochemical and morphological effects of temperature on Typha latifolia L. (Typhaceae) originating from different ends of a thermal gradient. Am J Bot 66:902–906
Jones ME (1971) The population genetics of Arabidopsis thaliana (L.) Heynh. I. The breeding system. II Population structure. III. The effect of vernalisation. Heredity 27:39–72
Liu EH, Sharitz RR, Smith MH (1978) Thermal sensitivities of malate dehydrogenase isozymes in Typha. Amer J Bot 65:214–220
McNaughton SJ (1972) Enzymic thermal adaptations. The evolution of homeostasis in plants. Amer Nat 106:165–172
McNaughton SJ (1974) Natural selection at the enzyme level. Amer Nat 108:616–624
Potvin C (1982) Adaptation et acclimatation à la temperature chez Arabidopsis thaliana (L.) Heynh. Unpublished MSC Thesis, Université de Montréal, Montréal 193
Potvin C, Simon J-P, Blanchard M-H (1983) Thermal properties of NAD malate dehydrogenase and glutamate oxaloacetate transaminase in two genotypes of Arabidopsis thaliana (Cruciferae) from contrasting environments. Plant Science Letters. (In Press)
Radcliffe D (1961) Adaptation in habitat in a group of annual plants. J Ecol 49:187–203
Redei GP (1970) Arabidopsis thaliana (L.) Heynh. A review of the genetics and biology. Bibliographia Genetica 20(2):1–151
Redei GP (1975) Arabidopsis as a genetic tool. Annu Rev Genet 9:111–128
Shcherbakova AM, Feldman NL (1982) Electrophoretic patterns and thermostability of some proteins from heat-hardened wheat. J Thermal Biol 7:111–116
Simon J-P (1979a) Differences in thermal properties of NAD dehydrogenase in gentotypes of Lathyrus japonicus Willd. (Leguminosae) from maritime and continental sites. Plant Cell Environ 2:23–33
Simon J-P (1979b) Adaptation and acclimation of higher plants at the enzyme level: latitudinal variations of thermal properties of NAD malate dehydrogenase in Lathyrus japonicus Willd. (Leguminosae). Oecologia (Berlin) 39:273–287
Simon J-P (1979c) Adaptation and acclimation of higher plants at the enzyme level: temperature-dependent substrate binding ability of NAD malate dehydrogenase in four populations of Lathyrus japonicus Willd. (Leguminosae). Plant Sci Lett 14:113–120
Simon J-P (1979d) Adaptation and acclimation of higher plants at the enzyme level: speed of acclimation for apparent energy of activation of NAD malate dehydrogenase in Lathyrus japonicus Willd. (Leguminosae). Plant Cell Environ 2:35–38
Snape JW, Lawrence MJ (1971) The breeding system of Arabidopsis thaliana. Heredity 27:299–302
Somero GN (1975) Temperature as a selective factor in protein evolution: The adaptational strategy of “compromise”. J Exp Zool 194:175–188
Somero GN (1978) Temperature adaptation of enzymes: Optimization through structure-function compromises. Ann Rev Ecol Syst 9:1–29
Somero GN, Low PS (1977) Eury-tolerant proteins: Mechanisms for extending the environmental tolerance range of enzyme-ligand interactions. Amer Nat 111:527–538
Teeri JA, Peet MM (1978) Adaptation of malate dehydrogenase to environmental temperature variability of two populations of Potentilla glandulosa Linde. Oecologia (Berlin) 34:133–141
Ting TP, Fuhr I, Curry R, Zschoche WC (1975) Malate dehydrogenase isozymes in plants: preparation, properties, and biological significance. In: Isozymes, vol. II. Physiological function Marker CL (ed). Academic Press, New York, pp 369–384
Van Chi-Bonnardel R (1973) The atlas of Africa. Editions Jeune Afrique, Paris
Vroman HE, Brown JRC (1963) The effect of temperature on the activity of succinic dehydrogenase from livers of rats and frogs. J Comp Physiol 61:129–131
Walter H, Lieth H (1964) Klimadiagramm Weltatlas. VEB Gustav Fischer Verlag, Jena
Wilkinson GN (1961) Statistical estimations in enzyme kinetics. J Biochem 80:324–332
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Simon, JP., Potvin, C. & Blanchard, MH. Thermal adaptation and acclimation of higher plants at the enzyme level: kinetic properties of NAD malate dehydrogenase and glutamate oxaloacetate transaminase in two genotypes of Arabidopsis thaliana (Brassicaceae). Oecologia 60, 143–148 (1983). https://doi.org/10.1007/BF00379515
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00379515