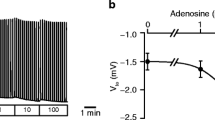
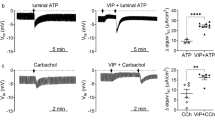
Abstract
Transepithelial solute transport and bicarbonate secretion are major functions of pancreatic duct cells, and both functions are thought to involve the presence of chloride channels in the apical membrane of the cell. After being isolated from a human pancreatic adenocarcinoma, the Capan-1 cell line conserves most of the properties of ductal cells and thus constitutes a useful system for investigating the role of chloride channels. Using patch-clamp techniques, we identified three different chloride-selective channels in the apical membrane of confluent Capan-1 cells. Two were non-rectifying chloride channels with low (50 pS) and high (350 pS) unitary conductances. Both channels were active in cell-attached recordings, and they were consistently located together in the same patch. Maxi Cl− channels displayed multiple subconductance states, and were reversibly inactivated by either positive or negative voltage changes, which indicates that they were optimally opened at the cell resting potential. The third was an outwardly rectifying chloride channel with a unitary conductance of 38 pS and 70 pS at negative and positive potentials respectively. Rectifying Cl− channels were clustered in discrete loci. They were silent in situ, but became active after patch excision. In inside-out excised patches, the three channels displayed a high selectivity for Cl− over monovalent cations (Na+ and K+) and gluconate. They were blocked by 20–200 μM 4,4′-diisothiocyanatostilbene-2,2′-disulfonic acid (DIDS) and were insensitive to changes in the Ca2+ concentration. Our results show that the apical membrane of Capan-1 cells contains a high density of chloride channels; these channels may provide pathways for transepithelial solute transport as well as for bicarbonate secretion.
Similar content being viewed by others
References
Case RM, Argent BE (1986) Bicarbonate secretion by pancreatic duct cells: mechanisms and control. In: Go VLW, et al. (eds) The exocrine pancreas: biology, pathobiology and diseases. Raven, New York, pp 213–243
Christensen O, Simon M, Randlev T (1989) Anion channels in a leaky epithelium. A patch-clamp study of choroid plexus. Pflügers Arch 415:37–46
Diener M, Rummel W, Mestres P, Lindemann B (1989) Single chloride channels in colon mucosa and isolated colonic enterocytes of the rat. J Membr Biol 108:21–30
Giraldez F, Murray KJ, Sepulveda FV, Sheppard DN (1989) Characterization of a phosphorylation-activated Cl− -selective channel in isolated Necturus enterocytes. J Physiol (Lond) 416:517–537
Gray MA, Greenwell JR, Argent BE (1988) Secretin-regulated chloride channel on the apical plasma membrane of pancreatic duct cells. J Membr Biol 105:131–142
Gray MA, Harris A, Coleman L, Greenwell JR, Argent BE (1989) Two types of chloride channel on duct cells cultured from human fetal pancreas. Am J Physiol 257:C240-D251
Gray MA, Pollard CE, Harris A, Coleman L, Greenwell JR, Argent BE (1990) Anion selectivity and block of the small conductance chloride channel on pancreatic duct cells. Am J Physiol 259:C752-C761.
Halm DR, Rechkemmer GR, Schoumacher RA, Frizzell RA (1988) Apical membrane chloride channels in a colonic cell line activated by secretory agonists. Am J Physiol 254:C505-C511
Hanrahan JW, Alles WP, Lewis SA (1985) Single anion-selective channels in basolateral membrane of a mammalian tight epithelium. Proc Natl Acad Sci USA 82:7791–7795
Hayslett JP, Gögelein H, Kunzelmann K, Greger R (1987) Characteristics of apical chloride channels in human colon cells (HT 29). Pflügers Arch 410:487–494
Jorissen M, Vereecke J, Carmeliet E, Van den Berghe H, Cassiman JJ (1990) Outward-rectifying chloride channels in cultured adult and fetal human nasal epithelial cells. J Membr Biol 117:123–130
Kolb HA, Brown CDA, Murer H (1985) Identification of a voltage-dependent anion channel in the apical membrane of a Cl−-secretory epithelium (MDCK). Pflügers Arch 403:262–265
Krouse ME, Schneider GT, Gage PW (1986) A large anion-selective channel has seven conductance levels. Nature 319:58–60
Krouse ME, Hagiwara G, Chen J, Lewiston NJ, Wine JJ (1989) Ion channels in normal human and cystic fibrosis sweat gland cells. Am J Physiol 257:C129-C140
Kunzelmann K, Pavenstädt H, Greger R (1989) Properties and regulation of chloride channels in cystic fibrosis and normal airway cells. Pflügers Arch 415:172–182
Kyriazis AP, Kyriazis AA, Scarpelli DG, Fogh J, Rao MS, Lepera R (1982) Human pancreatic adenocarcinoma line CAPAN 1 in tissue culture and the nude mouse. Am J Pathol 106:250–260
Levrat JH, Palevody C, Daumas M, Ratovo G, Hollande E (1988) Differentiation of the human pancreatic adenocarcinoma cell line (Capan 1) in culture and co-culture with fibroblasts: dome formation. Int J Cancer 42:615–621
Oberleithner H, Vogel U, Kersting U (1990) Madin-Darby canine kidney cells. I. Aldosterone-induced domes and their evaluation as a model system. Pflügers Arch 416:526–532
Ruellan C, Scemama JL, Clerc P, Fagot-Revurat P, Clemente F, Ribet A (1986) VIP regulation of a human pancreatic cancer cell line: Capan 1. Peptides 7 [Suppl 1] 267–271
Schoumacher RA, Ram J, Lannuzzi MC, Bradburry NA, Wallace RW, Tom Hon C, Kelly DR, Schmid SM, Gelder FB, Rado TA, Frizzel RA (1990) A cystic fibrosis pancreatic adenocarcinoma cell line. Proc Natl Acad Sci USA 87:4012–4016
Soejima M, Kokubun S (1988) Single anion-selective channel and its ion selectivity in the vascular smooth muscle cell. Pflügers Arch 411:304–311
Tabcharani JA, Jensen TJ, Riordan JR, Hanrahan JW (1989) Bicarbonate permeability of the outwardly rectifying anion channel. J Membr Biol 112:109–122
Velasco G, Prieto M, Alvarez-Riera J, Gascon S, Barros F (1989) Characteristics and regulation of a high conductance anion channel in GBK kidney epithelial cells. Pflügers Arch 414:304–310
Welsh MJ (1990) Abnormal regulation of ion channels in cystic fibrosis epithelia. FASEB J 4:2718–2725
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Becq, F., Fanjul, M., Mahieu, I. et al. Anion channels in a human pancreatic cancer cell line (Capan-1) of ductal origin. Pflugers Arch. 420, 46–53 (1992). https://doi.org/10.1007/BF00378640
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00378640