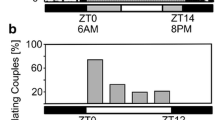
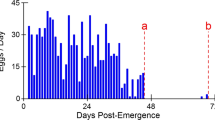
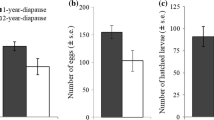
Summary
Flesh flies (Sarcophagidae) collected in Costa Rica and Panama lack the pupal diapause that is characteristic of flesh flies from the temperate zone and tropical Africa. The absence of a diapause capacity in the neotropical species correlates with several other life history traits: in most species the post feeding wandering phase of the third larval instar is longer and duration is more variable, adult life is longer, clutch size is smaller, and more clutches are produced. Among species that have the capacity for diapause, risk is invested primarily in the diapausing stage and other life stages are brief. Though diapausing species are short-lived, they produce as many or more progeny than nondiapausing species by increasing clutch size. The slower and more variable developmental rate and increased adult longevity desynchronizes development and permits the nondiapausing species to spread an environmental risk over different stages of the life cycle, thus offering an alternative to diapause. Other traits such as body size, developmental velocity, thermal constant thresholds, thermal constants, age at first reproduction, and the interval between clutches do not appear related to the capacity for diapause.
Similar content being viewed by others
References
Chen CP, Denlinger DL, Lee RJ Jr (1987) Responses of nondiapausing flesh flies (Diptera: Sarcophagidae) to low rearing temperatures: developmental rate, cold tolerance and glycerol concentrations. Ann Entomol Soc Am 80:790–796
Denlinger DL (1972a) Induction and termination of pupal diapause in Sarcophaga flesh flies. Biol Bull 142:11–24
Denlinger DL (1972b) Seasonal phenology of diapause in the flesh fly Sarcophaga bullata. Ann Entomol Soc Am 65:410–414
Denlinger DL (1974) Diapause potential in tropical flesh flies. Nature 252:223–224
Denlinger DL (1978) The developmental response of flesh flies (Diptera: Sarcophagidae) to tropical seasons: variation in generation time and diapause in East Africa. Oecologia (Berlin) 35:105–107
Denlinger DL (1979) Pupal diapause in tropical flesh flies: environmental and endocrine regulation, metabolic rate and genetic selection. Biol Bull 156:31–46
Denlinger DL (1981a) The physiology of pupal diapause in flesh flies. In: Bhaskaran G, Friedman S, Rodriguez JG (eds) Current topics in insect endocrinology and nutrition. Plenum Press, New York, pp 131–160
Denlinger DL (1981b) Basis for a skewed sex ratio in diapause-destined flesh flies. Evolution 35:1247–1248
Denlinger DL (1986) Dormancy in tropical insects. Annu Rev Entomol 31:239–264
Denlinger DL, Shukla M (1984) Increased length and variability of the life cycle in tropical flesh flies (Diptera: Sarcophagidae) that lack pupal diapause. Ann Entomol Soc Am 77:46–49
Deseo KV (1973) Side-effect of diapause inducing factors on the reproductive activity of some lepidopterous species. Nature New Biol 242:126–127
Dietrich WE, Windsor DM, Dunne T (1982) Geology, climate, and hydrology of Barro Colorado Island. In: Leigh EG Jr, Rand SA, Windsor DM (eds) The ecology of a tropical forest. Smithsonian, Washington, D.C., pp. 21–46
Dingle H (1981) Geographic variation and behavioral flexibility in milkweed bug life histories. In: Denno RF, Dingle H (eds) Insect life history patterns. Springer, New York Heidelberg Berlin. pp. 57–73
Dingle H, Alden BM, Blakley NR, Kopec D, Miller ER (1980) Variation in photoperiodic response within and among species of milkweed bugs (Oncopeltus). Evolution 34:356–370
Downes WL Jr (1965) Family Sarcophagidae. In: Stone A, Sabrosky CW, Wirth WW, Foote RH, Coulson JR (eds) Catalog of the Diptera of America North of Mexico. U.S. Govt. Printing Office, Washington, D.C., pp. 933–961
Fraenkel G, Hsiao C (1968) Manifestations of a pupal diapause in two species of flies, Sarcophaga argyrostoma and S. bullata. J Insect Physiol 14:689–705
Henrich VC, Denlinger DL (1982) Selection for late pupariation affects diapause incidence and duration in the flesh fly, Sarcophaga bullata. Physiol Entomol 7:407–411
Hoy MA (1978) Variability in diapause attributes of insects and mites: some evolutionary and practical implications. In: Dingle H (ed) Evolution of insect migration and diapause. Springer, New York Heidelberg Berlin, pp. 101–126
Laudien H (1973) Changing reaction systems. In: Precht H, Christophersen J, Hensel H, Larcher W (eds) Temperature and life. Springer, New York Heidelberg Berlin, pp. 355–399
Moeur JE, Istock CA (1980) Ecology and evolution of the pitcherplant mosquito. IV. Larval influence over adult reproductive performance and longevity. J Anim Ecol 49:775–792
Rohdendorf BB (1967) The trends of historical development of the Sarcophagidae (In Russian). Proc Entomol Inst Acad Sci USSR 116:1–91
Saunders DS (1971) The temperature-compensated photoperiodic clock ‘programming’ development and pupal diapause in the flesh-fly, Sarcophaga argyrsotoma. J Insect Physiol 17:801–812
Tanaka S, Wolda H, Denlinger DL (1987) Seasonality and its physiological regulation in three neotropical insect taxa from Barro Colorado Island, Panama. Insect Sci Appl 8:507–514
Tauber MJ, Tauber CA, Masaki S (1986) Seasonal adaptations of insects. Oxford University Press, Oxford
Waldbauer GP (1978) Phenological adaptation and the polymodal emergence patterns of insects. In: Dingle H (ed) Evolution of insect migration and diapause. Springer, New York Heidelberg Berlin, pp. 127–144
Zumpt F (1972) Calliphoridae (Diptera Cyclorrapha) Part IV: Sarcophagidae. Parc Nat Virunga-Misson GF De Witte (1933–1935) 101:1–264
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Denlinger, D.L., Chen, C.P. & Tanaka, S. The impact of diapause on the evolution of other life history traits in flesh flies. Oecologia 77, 350–356 (1988). https://doi.org/10.1007/BF00378041
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00378041