Abstract
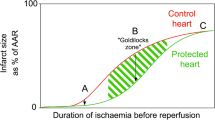
An important mechanism of lethal myocardial reperfusion injury is the development of cellular hypercontracture at the onset of reperfusion. Hypercontracture can lead to cytolysis by mutual mechanical disruption of myocardial cells. 2,3-Butanedione monoxime (BDM) inhibits myofibrillar cross-bridge cycling and may therefore reduce infarct size in ischaemic reperfused myocardium. This study investigated whether a temporary presence of BDM protects against myocardial reperfusion injury in an intact-animal preparation. Anaesthetized open-chest dogs (n=10) underwent 1 h of left anterior descendent artery (LAD) occlusion and received intracoronary BDM (25 mM, n=5) or vehicle (n=5) for 65 min starting with an anoxic local infusion 5 min before reperfusion. Infarct size was assessed by triphenyltetrazolium staining after 6 h reperfusion. The infusion of BDM was accompanied by a transient reduction of left ventricular systolic pressure from 84.3±11.2 mm Hg during occlusion to 66.4±9.9 mm Hg at 30 min reperfusion (mean±SD, P<0.01 vs. control). LAD-flow and regional wall motion in the area at risk showed no difference between groups. Infarct size (% of area at risk) was reduced from 24.4±8.7 (control) to 6.6±2.0% (BDM) (P<0.01). The results demonstrate that development of necrosis in reperfused myocardium can be greatly reduced by temporary presence of the contractile inhibitor BDM at the onset of reperfusion.
Similar content being viewed by others
References
Axelrod HI, Galloway AC, Murphy MS, Laschinger JC, Grossi EA, Baumarm G, Colvin SB, Hunter CE, Glassman E, Spencer FC (1987) A comparison of methods for limiting myocardial infarct expansion during acute reperfusion — primary role of unloading. Circulation 76 (suppl 5): V28-V32
Blanchard EM, Smith GL, Allen DG, Alpert NR (1990) The effects of 2,3-butanedione monoxime on initial heat, tension and aequorin light output on ferret papillary muscles. Pflügers Arch 416: 219–221
Elz JS, Nayler WG (1988) Contractile activity and reperfusion-induced calcium gain after ischemia in the isolated rat heart. Lab Invest 58: 653–659
Fryer MF, Neering IR, Stephenson DG (1988) Effects of 2,3-butanedione monoxime on the contractile activation of fast- and slow switch rat muscle fibres. J Physiol (Lond) 407: 53–75
Gallagher KP (1990) Regional myocardial flow-function relationship in ischemia. In: Heusch G (ed) Pathophysiology and rational pharmacotherapy of myocardial ischemia. Steinkopff, Darmstadt, pp 111–135
Ganote CE (1983) Contraction band necrosis and irreversible myocardial injury. J Mol Cell Cardiol 15: 67–73
Ganote CE, Sims MD, Safavi S (1982) Effects of dimethylsulfoxide (DMSO) on the oxygen paradox in perfused rat hearts. Am J Path 109: 270–276
Garcia-Dorado D, Théroux P, Duran JM, Solares J, Alonso J, Sanz E, Munoz R, Elizaga J, Botas J, Fernandez-Avilés F, Soriano J, Esteban E (1992) Selective inhibition of the contractile apparatus: a new approach to modification of infarct size, infarct composition, and infarct geometry during coronary artery occlusion and reperfusion. Circulation 85: 1160–1174
Gwathmey JK, Hajjar RJ, Solaro RJ (1991) Contractile deactivation and uncoupling of crossbridges: effects of 2,3-butanedione monoxime on mammalian myocardium. Circ Res 69: 1280–1292
Hearse DJ, Humphrey SM, Chain EB (1973) Abrupt reoxygenation of the anoxic potassium-arrested perfused rat heart: A study of myocardial enzyme release. J Mol Cell Cardiol 5:395–407
Klein HH, Puschmann S, Schaper J, Schaper W (1981) The mechanism of the tetrazolium reaction in identifying experimental myocardial infarction. Virchows Arch [A] 393: 287–297
Li T, Sperelakis N, TenEick RE, Solaro JR (1985) Effects of diacetyl monoxime on cardiac excitation-contraction coupling. J Pharmacol Exp Ther 232: 688–695
Liu Y, Cabo C, Salomonsz R, Delmar M, Davidenko J, Jalife J (1993) Effects of diacetyl monoxime on the electrical properties of sheep and guinea pig ventricular muscle. Cardiovasc Res 27: 1991–1997
Piper HM (1990) Mitochondrial injury in the oxygen-depleted and reoxygenated myocardial cell. In: Piper HM (ed) Pathophysiology of severe ischemic injury. Kluwer, Dordrecht, pp 91–114
Rosenkranz ER, Buckberg GD (1983) Myocardial protection during surgical coronary reperfusion. J Am Coll Cardiol 1: 1235–1246
Schipke J, Heusch G, Schulz R, Thämer V (1987) An easy and quick implantation procedure for the measurement of myocardial wall thickness using sonomicrometry. Basic Res Cardiol 82: 411–414
Schülter KD, Schwartz P, Siegmund B, Piper HM (1991) Prevention of the oxygen paradox in hypoxic-reoxygenated hearts. Am J Physiol 261: H416-H432
Siegmund B, Koop A, Klietz T, Schwartz P, Piper HM (1990) Sarcolemmal integrity and metabolic competence of cardiomyocytes under anoxia-reoxygenation. Am J Physiol 258: H285-H291
Siegmund B, Klietz T, Schwartz P, Piper HM (1992) Temporary contractile blockade prevents hypercontracture in anoxicreoxygenated cardiomyocytes. Am J Physiol 260:H426-H435
Siegmund B, Zude R, Piper HM (1992) Recovery of anoxicreoxygenated cardiomyocytes from severe Ca2+ overload. Am J Physiol 263: H1262-H1269
Siegmund B, Schlüter KD, Piper HM (1993) Calcium and the oxygen paradox. Cardiovasc Res 27: 1778–1783
Steele DS, Smith GL (1993) Effects of 2,3-butanedione monoxime on sarcoplasmic reticulum of saponin-treated rat cardiac muscle. Am J Physiol 265: H1493-H1500
Vivaldi MT, Kloner RA, Schoen FJ (1985) Triphenyltetrazolium staining of irreversible ischemic injury following coronary artery occlusion in rats. Am J Pathol 121: 522–530
West JM, Stephenson DG (1989) Contractile activation and the effects of 2,3-butanedione monoxime (BDM) in skinned cardiac preparations from normal and dystrophic mice (129/ReJ). Pflügers Arch 413: 546–552
Winkle DM van, Matsuki T, Gad NM, Jordan MC, Downey JM (1990) Left ventricular unloading during reperfusion does not limit myocardial infarct size. Circulation 81: 1374–1379
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schlack, W., Uebing, A., Schäfer, M. et al. Regional contractile blockade at the onset of reperfusion reduces infarct size in the dog heart. Pflügers Arch 428, 134–141 (1994). https://doi.org/10.1007/BF00374850
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374850