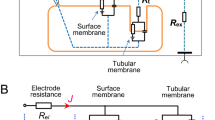
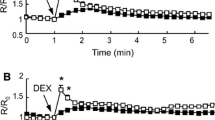
Abstract
Rat hepatocytes in primary culture were impaled with conventional microelectrodes. Addition of 5–100 μmol/l taurocholate led to a slowly developing depolarization that was maximal at 50 μmol/l (10.5±1.5 mV, n=15) and not reversible. The effect was Na+ dependent and decreased in cells preincubated with 1 μmol/l taurocholate. Increasing external K+ tenfold depolarized the cells by 12.3±2.3 mV under control conditions and by 6.3±1.2 mV with 50 μmol/l taurocholate present (n=7). Depolarization by 1 mmol/l Ba2+ was 7.6±0.8 mV and 6.0±0.7 mV (n=9) before and after addition of taurocholate, respectively. Cable analysis and Na+ substitution experiments reveal that this apparent decrease in K+ conductance reflects an actual increase in Na+ conductance: in the presence of taurocholate, specific cell membrane resistance decreased from 2.8 to 2.3 kΩ · cm2 · Na+ substitution by 95% depolarized cell membranes by 8.9±2.9 mV (n=9), probably due to indirect effects on K+ conductance via changes in cell pH. With taurocholate present, the same manoeuvre changed membrane voltages by −0.8±2.6 mV. When Na+ concentration was restored to 100% from solutions containing 5% Na+, cells hyperpolarized by 3.5±3.6 mV (n=7) under control conditions and depolarized by 4.4±2.9 mV in the presence of taurocholate, respectively. In Cl− substitution experiments, there was no evidence for changes in Cl− conductance by taurocholate. These results show that taurocholate-induced membrane depolarization is due to an increase in Na+ conductance probably via uptake of the bile acid.
Similar content being viewed by others
References
Aldini R, Roda A, Lenzi P, Calcaterra D, Vaccari C, Calzolari M, Festi D, Mazzella G, Bazzoli F, Roda E, Forti GC (1992) Hepatic uptake and biliary secretion of bile acids in the perfused rat liver. Pharmacol Rev 25:51–61
Bear CE, Davison JS, Shaffer EA (1987) Sodium-dependent taurocholate uptake by isolated rat hepatocytes occurs through an electrogenic mechanism. Biochim Biophys Acta 903:388–394
Bear CE, Davison JS, Shaffer EA (1988) Intracellular pH influences the resting membrane potential of isolated rat hepatocytes. Biochim Biophys Acta 944:113–120
Benedetti A, Strazzabosco M, Corasanti JG, Haddad P, Graf J, Boyer JL (1991) C1−-HCO −3 exchanger in isolated rat hepatocytes: role in regulation of intracellular pH. Am J Physiol 261:G 512-G 522
Blitzer BL, Donovan CB (1984) A new method for the rapid isolation of basolateral plasma membrane vesicles from rat liver. Characterization, validation, and bile acid transport studies. J Biol Chem 259:9295–9301
Blitzer BL, Ratoosh SL, Donovan CB, Boyer JL (1982) Effects of inhibitors of Na+-coupled ion transport on bile acid uptake by isolated rat hepatocytes. Am J Physiol 243:G 48-G 53
Edmondson JW, Miller BA, Lumeng L (1985) Effect of glucagon on hepatic taurocholate uptake: relationship to membrane potential. Am J Physiol 249:G 427-G 433
Fitz JG, Scharschmidt BF (1987) Regulation of transmembrane electrical potential gradient in rat hepatocytes in situ. Am J Physiol 252:G 56-G 64
Fitz JG, Persico M, Scharschmidt BF (1989) Electrophysiological evidence for Na+-coupled bicarbonate transport in cultured rat hepatocytes. Am J Physiol 256:G 491-G 500
Fitz JG, Trouillot TE, Scharschmidt BF (1989) Effect of pH on membrane potential and K+ conductance in cultured rat hepatocytes. Am J Physiol 257:G 961-G 968
Frömter E (1972) The route of passive ion movement through the epithelium of Necturus gallbladder. J Membr Biol 8:259–301
Hall TJ, Baker AL, Cooper MJ, Moossa AR (1979) Choleresis and cholestasis produced by infusion of taurocholic acid or taurodehydrocholic acid combined with BSP in the rhesus monkey. Dig Dis Sci 24:351–357
Hardison WGM, Hatoff DE, Miyai K, Weiner RG (1981) Nature of bile acid maximum secretory rate in the rat. Am J Physiol 241:G 337-G 343
Henderson RM, Graf J, Boyer JL (1987) Na-H exchange regulates intracellular pH in isolated rat hepatocyte couplets. Am J Physiol 252:G 109-G 113
Herz R, Paumgartner G, Preisig R (1976) Inhibition of bile formation by high doses of taurocholate in the perfused rat liver. Scand J Gastroenterol 11:741–746
Inoue M, Kinne R, Tran T, Arias IM (1982) Taurocholate transport by rat liver sinusoidal membrane vesicles:Evidence of sodium cotransport. Hepatology 2:572–579
Lidofsky SD, Scharschmidt BF (1992) Hepatocellular taurocholate uptake is electrogenic and regulated by transmembrane electrical potential difference. Gastroenterology 102:A842
Petzinger E, Müller N, Föllmann W, Deutscher H, Kinne RKH (1989) Uptake of bumetanide into isolated rat hepatocytes and primary liver cell cultures. Am J Physiol 256:G 78-G 86
Reichen J, Paumgartner G (1976) Uptake of bile acids by perfused rat liver. Am J Physiol 231:734–742
Ruifrok PG, Meijer DKF (1982) Sodium ion-coupled uptake of taurocholate by rat-liver plasma membrane vesicles. Liver 2:28–34
Scharschmidt BF, Stephens JE (1981) Transport of sodium, chloride, and taurocholate by cultured rat hepatocytes. Proc Natl Acad Sci USA 78:986–990
Schwarz LR, Burr R, Schwenk M, Pfaff E, Greim H (1975) Uptake of taurocholic acid into isolated rat-liver cells. Eur J Biochem 55:617–623
Wehner F, Guth D (1991) Electric properties of rat liver cell cultures on gas-permeable membranes. Am J Physiol 261:G 407-G 416
Wehner F, Mooren F, Kinne RKH (1991) Taurocholate depolarizes membrane voltage of rat hepatocytes in primary culture. Pflügers Arch 418:R71
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wehner, F. Taurocholate depolarizes rat hepatocytes in primary culture by increasing cell membrane Na+ conductance. Pflugers Arch. 424, 145–151 (1993). https://doi.org/10.1007/BF00374605
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374605