Abstract
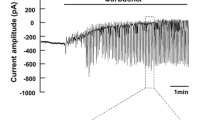
We have previously shown that mouse mandibular granular ducts contain a hyperpolarization-activated Cl− conductance. We now show that the instantaneous current/voltage (I/V) relation of this Cl− conductance is inwardly rectifying with a slope conductance of 15.4±1.8 nS (n=4) at negative potentials and of 6.7±0.9 nS (n=4) at positive potentials. Thus, the inward rectification seen in the steady-state I/V relation is due, not only to voltage activation of the Cl− conductance, but also to the intrinsic conductance properties of the channel. We show further that the ductal Cl− conductance is not activated by including ATP (10 mmol/l) in the pipette solution. Finally, we show that the conductance is not blocked by the addition of any of the following compounds to the extracellular solution: anthracene-9-carboxylate (A9C, 1 mmol/l), diphenylamine-2-carboxylate (DPC, 1 mmol/l), 5-nitro-2-(3-phenylpropylamino)-benzoate (NPPB, 100 μmol/l), 4,4′-diisothiocyanato-stilbene-2,2′-disulphonate (DIDS, 100 μmol/l), indanyloxyacetic acid (IAA-94, 100 μmol/l), verapamil (100 μmol/l), glibenclamide (100 μmol/l) and Ba2+ (5 mmol/l). The properties of the ductal Cl− conductance most nearly resemble those of the ClC-2 channel. Both channel types have instantaneous I/V relations that are slightly inwardly rectifying, are activated by hyperpolarization with a time-course in the order of hundreds of milliseconds, have a selectivity sequence of Br−>Cl−>I−, and are insensitive to DIDS. The only identified difference between the two is that the ClC-2 channel is 50% blocked both by DPC and A9C (1 mmol/l), whereas the ductal Cl− conductance is insensitive to these compounds.
Similar content being viewed by others
References
Anderson MP, Welsh MJ (1991) Calcium and cAMP activate different chloride channels in the apical membrane of normal and cystic fibrosis epithelia. Proc Natl Acad Sci USA 88:6003–6007
Anderson MP, Berger HA, Rich DP, Gregory RJ, Smith AE, Welsh MJ (1991) Nucleoside triphosphates are required to open the CFTR chloride channel. Cell 67:775–784
Anderson MP, Sheppard DN, Berger HA, Welsh MJ (1992) Chloride channels in the apical membrane of normal and cystic fibrosis airway and intestinal epithelia. Am J Physiol 263:L1-L14
Attali B, Guillemare E, Lesage F, Honoré E, Romey G, Lazdunski M, Barhanin J (1993) The protein IsK is a dual activator of K+ and Cl− channels. Nature 365:850–852
Augustus J, Bijman J, Van Os CH (1978) Electrical resistance of rabbit submaxillary main duct: a tight epithelium with leaky cell membranes. J Membr Biol 43:203–226
Barry PH, Lynch JW (1991) Liquid junction potentials and small cell effects in patch clamp analysis. J Membr Biol 121:101–117
Bijman J (1982) Transport parameters of the main duct of the rabbit mandibular salivary gland. Dissertation, University of Nijmegen, Nijmegen
Cook DI, Gard GB, Champion M, Young JA (1988) Patchclamp studies of the electrolyte secretory mechanism of rat mandibular gland cells stimulated with acetylcholine or isoproterenol. In: Thorn NA, Treiman M, Petersen OH, Thaysen JH (eds) Molecular mechanisms in secretion (25th Alfred Benzon Symposium 1987). Munksgaard, Copenhagen, pp 131–151
Dinudom A, Young JA, Cook DI (1993) Amiloride-sensitive Na+ current in the granular duct cells of mouse mandibular glands. Pflügers Arch 423:164–166
Dinudom A, Young JA, Cook DI (1993) Na+ and Cl− conductances are controlled by cytosolic Cl− concentration in the intralobular duct cells of mouse mandibular glands. J Membr Biol 135:289–295
Dinudom A, Young JA, Cook DI (1994) Ion channels in the basolateral membrane of intralobular duct cells of mouse mandibular glands. Pflügers Arch 428:202–208
Evans MG, Marty A (1986) Calcium-dependent chloride currents in isolated cells from rat lacrimal glands. J Physiol (Lond) 378:437–460
Evans MG, Marty A, Tan YP, Trautmann A (1986) Blockage of Ca-activated Cl conductance by furosemide in rat lacrimal glands. Pflügers Arch 406:65–68
Frömter E, Gebler B, Schopow K, Pockrandt-Hemstedt H (1974) Cation and anion permeability of rabbit submaxillary main duct. In: Thorn NA, Petersen OH (eds) Secretory mechanisms of exocrine glands. Munksgaard, Copenhagen, pp 496–513
Gründer S, Thiemann A, Pusch M, Jentsch TJ (1992) Regions involved in the opening of ClC-2 chloride channel by voltage and cell volume. Nature 360:759–762
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FS (1981) Improved patch-clamp techniques for high resolution current recording from cells and cell free membrane patches. Pflügers Arch 391:85–100
Hayslett JP, Gögelein H, Kunzelmann K, Greger R (1987) Characteristics of apical chloride channels in human colon cells (HT29). Pflügers Arch 410:487–494
Ishikawa T, Cook DI (1993) A Ca2+-activated Cl− current in sheep parotid secretory cells. J Membr Biol 135:261–271
Ishikawa T, Cook DI (1994) Characterization of an outwardly rectifying chloride channel in a human submandibular gland duct cell line (HSG). Pflügers Arch 427:203–209
Kunzelmann K, Pavenstädt H, Greger R (1989) Properties and regulation of chloride channels in cystic fibrosis and normal airway cells. Pflügers Arch 415:172–182
Martin DK (1993) Small conductance chloride channels in acinar cells from the rat mandibular salivary gland are directly controlled by a G protein. Biochem Biophys Res Commun 192:1266–1273
Marty A, Tan YP, Trautmann A (1984) Three types of calcium-dependent channels in rat lacrimal glands. J Physiol (Lond) 357:293–325
Melvin JE, Kawaguchi M, Baum BJ, Turner RJ (1987) A muscarinic agonist-stimulated chloride efflux pathway is associated with fluid secretion in rat parotid acinar cells. Biochem Biophys Res Commun 145:754–759
Moorman JR, Palmer CJ, John JE, Durieux ME, Jones LR (1992) Phospholemman expression induces a hyperpolarization-activated chloride current in Xenopus oocytes. J Biol Chem 267:14551–14554
Paulmichl M, Li Y, Wickman K, Ackerman M, Peralta E, Clapham D (1992) New mammalian chloride channel identified by expression cloning. Nature 356:238–241
Quinton PM, Reddy MM (1992) Control of CFTR chloride conductance by ATP levels through nonhydrolytic binding. Nature 360:79–81
Sheppard DN, Welsh MJ (1992) Effect of ATP sensitive K+ channel regulators on cystic fibrosis transmembrane conductance regulator chloride currents. J Gen Physiol 100:573–591
Slegers JFG, Moons WM, Idzerda PP, Stadhouders WM (1975) The contribution of a Cl-shunt to the transmucosal potential of the rabbit submaxillary duct. J Membr Biol 25:213–236
Sugimoto T, Tanabe Y, Shigemoto R, Iwai M, Takumi T, Ohkubo H, Nakanishi S (1990) Immunohistochemical study of a rat membrane protein which induces a selective potassium permeation: its localization in the apical membrane portion of epithelial cells. J Membr Biol 113:39–47
Thiemann A, Gründer S, Pusch M, Jentsch TJ (1992) A chloride channel widely expressed in epithelial and non-epithelial cells. Nature 356:57–60
Tilmann M, Kunzelmann K, Fröbe U, Cabantchik I, Lang HJ, Englert HC, Greger R (1991) Different types of blockers of the intermediate conductance outwardly rectifying chloride channel in epithelia. Pflügers Arch 418:556–563
Trezise AEO, Buchwald M (1991) In vivo cell specific expression of the cystic fibrosis transmembrane conductance regulator. Nature 353:434–437
Valverde MA, Diaz M, Sepulveda FV, Gill DR, Hyde SC, Higgins CF (1992) Volume-regulated chloride channels associated with the human multidrug-resistance P-glycoprotein. Nature 355:830–833
Wangemann P, Wittner M, Di Stefano A, Lang HJ, Schlatter E, Greger R (1986) Cl−-channel blockers in the thick ascending limb of the loop of Henle. Structure activity relationship. Pflügers Arch 407 [Suppl 2]:S128-S141
Young JA, Cook DI, Van Lennep EW, Roberts ML (1987) Secretion by the major salivary glands. In: Johnson L, Christensen J, Jackson M, Jacobson E, Walsh J (eds) Physiology of the gastrointestinal tract, 2nd edn, vol 2. Raven, New York, pp 773–815
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Komwatana, P., Dinudom, A., Young, J.A. et al. Characterization of the Cl− conductance in the granular duct cells of mouse mandibular glands. Pflugers Arch. 428, 641–647 (1994). https://doi.org/10.1007/BF00374588
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374588