Summary
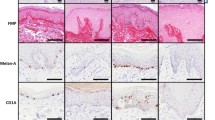
Recent biochemical and morphological investigations have provided evidence for a heterogeneous composition of keratohyalin in human skin. A major component is filaggrin. In interfollicular epidermis the heterogeneity of keratohyalin is not directly visible, whereas in normal ridged skin bicomponent keratohyalin is revealed by electron microscopy. Skin biopsies of ridged and non-ridged skin of normal individuals and patients with autosomal dominant ichthyosis vulgaris (ADI) — characterized by defective keratohyalin synthesis and lack of filaggrin — were investigated by routine transmission electron microscopy and immunogold postembedding techniques using a commercial monoclonal anti-filaggrin antibody. In normal interfollicular epidermis filaggrin labelling was demonstrated on keratohyalin granules and in the lowermost cornified cells, whereas in ADI patients crumbly keratohyalin granules were present that did not show specific labelling for filaggrin. In normal ridged skin only the major (more electron-dense) component reacted with anti-filaggrin, whereas the attached (less electron-dense) component did not react. Ridged skin of ADI patients contained globular keratohyalin that did not react with anti-filaggrin, thus corresponding to the attached keratohyalin component in normal ridged skin. Our results provide a visible counter-part to the recent biochemical investigations of keratohyalin protein heterogeneity and contribute to the understanding of terminal differentiation in human skin and of the defective keratohyalin synthesis in ADI.
Similar content being viewed by others
References
Allen MH, Tidman MJ, MacDonald DM (1986) Immunogold-silver staining in the localisation of cutaneous antigens. J Cutan Pathol 13:433
Allen MH, Tidman MJ, MacDonald DM (1988) Localization of cutaneous antigens by the immunogold-silver staining technique. Br J Dermatol 118:645–650
Anton-Lamprecht I (1973) Zur Ultrastruktur hereditÄrer Verhornungsstörungen. III. Autosomal-dominante Ichthyosis vulgaris. Arch Dermatol Forsch 248:149–172
Anton-Lamprecht I (1980) Keratinization of the human sweat duct. J Cutan Pathol 7:192–193
Anton-Lamprecht I (1981) Disturbances of tonofilament and keratohyalin structure and arrangement in inborn errors of keratinization. In: Marks R, Christophers E (eds) The epidermis in disease. MTP Press, Lancaster, pp 61–77
Anton-Lamprecht I (1982) New aspects of inherited ichthyoses and acrokeratoses as obtained from electron microscopy. In: Kukita A, Sciji M (eds) Proceedings of the XVIth International Congress of Dermatology, 23–28 May, Tokyo. University of Tokyo Press, Tokyo, pp 430–434
Anton-Lamprecht I (1983) Genetically induced abnormalities of epidermal differentiation and ultrastructure in ichthyoses and epidermolyses: pathogenesis, heterogeneity, fetal manifestation, and prenatal diagnosis. J Invest Dermatol 81:149s-156s
Anton-Lamprecht I, Schnyder UW (1974) Ultrastructure of inborn errors of keratinization. VI. Inherited ichthyoses — a model system for heterogeneities in keratinization disturbances. Arch Dermatol Forsch 250:207–227
Dale BA (1985) Filaggrin, the matrix protein of keratin. Am J Dermatopathol 7:65–68
Dale BA, Ling SY (1979) Immunologie cross-reaction of stratum corneum basic protein and a keratohyalin granule protein. J Invest Dermatol 72:257–261
Dale BA, Gown AM, Fleckman P, Kimball JR, Resing KA (1987) Characterization of two monoclonal antibodies to human epidermal keratohyalin: reactivity with filaggrin and related proteins. J Invest Dermatol 88:306–313
Fartasch M, Haneke E, Anton-Lamprecht I (1987) Ultrastructural study of the occurrence of autosomal dominant ichthyosis vulgaris in atopic eczema. Arch Dermatol Res 279:270–272
Fartasch M, Diepgen TL, Hornstein OP (1989) Are hyperlinear palms and dry skin signs of a concomitant autosomal dominant ichthyosis vulgaris in atopic dermatitis? Acta Derm Venereol (Stockh) [Suppl] 144:143–145
Fukuyama K, Epstein WL (1969) Sulfur-containing proteins and epidermal keratinization. J Cell Biol 40:830–838
Fukuyama K, Wier KA, Epstein WL (1972) Dense homogeneous deposits of keratohyaline granules in newborn rat epidermis. J Ultrastruct Res 38:16–26
Goldsmith LA (1976) The ichthyoses. Prog Med Genet 1:185–210
Haydok PV, Dale BA (1986) The repetitive structure of the profilaggrin gene as demonstrated using epidermal profilaggrin cDNA. J Biol Chem 261:12520–12525
Hohl D (1990) Ein neuer Stern am Himmel der epidermalen Proteine: Loricrin — was ist das? Hautarzt 41:299–301
Hohl D, Mehrel T, Lichti U, Yuspa S, Roop D, Steinert P (1989) Loricrin — the human glycine-rich cornified envelope precursor — isolation and characterization of a cDNA clone encoding a major component of the human cornified envelope. J Invest Dermatol 92:445
Holgate CS, Jackson P, Cown PN, Bird CC (1983) Immunogold-silver staining: new method of immunostaining with inhanced sensitivity. J Histochem Cytochem 31:938–944
Hoober JK, Bernstein IA (1966) Protein synthesis related to epidermal differentiation. Proc Natl Acad Sci USA 56:594–601
Jessen H (1970) Two types of keratohyaline granules. J Ultrastruct Res 33:95–115
Kastl I (1989) Zur normalen und pathologischen Keratinisierung der Leistenhaut (Palmae und Plantae). Charakterisierung und Abgrenzung einer neuen Palmoplantarkeratose mit autosomal-rezessivem Erbgang. Thesis, Faculty of Clinical Medicine, University of Heidelberg, FRG
Kastl I, Anton-Lamprecht I (1990) Bicomponent keratohyalin in normal kuman ridged skin. Arch Dermatol Res 282:71–75
Laurent R, Prost O, Nicollier M, Coumes Marquet S, Balzer MM, Adessi G (1985) Composite keratohyaline granules in palmoplantar keratoderma: an ultrastructural study. Arch Dermatol Res 277:384–394
Lonsdale-Eccles JD, Teller DC, Dale BA (1982) Characterization of a phosphorylated form of the intermediate filament-aggregating protein filaggrin. Biochemistry 21:5940–5948
Lonsdale-Eccles JD, Resing KA, Meek RL, Dale BA (1984) High-molecular-weight precursor of epidermal filaggrin and hypothesis for its tandem repeating structure. Biochemistry 23:1239–1245
Lynley AM, Dale BA (1983) The characterization of human epidermal filaggrin, a histidine-rich, keratin filament-aggregating protein. Biochim Biophys Acta 744:28–35
Matoltsy AG, Matoltsy MN (1966) The membrane protein of horny cells. J Invest Dermatol 46:127–129
Matoltsy AG, Matoltsy MN (1970) The chemical nature of keratohyaline granules of the epidermis. J Cell Biol 47:593–603
Matoltsy AG, Lavker RM, Matoltsy MN (1974) Demonstration of cystine-containing protein in keratohyalin granules of the epidermis. J Invest Dermatol 62:406–410
McKinley Grant LJ, Idler WW, Bernstein IA, Parry DA, Cannizzaro L, Croce CM, Huebner K, Lessin SR, Steinert PM (1989) Characterization of a cDNA clone encoding human filaggrin and localization of the gene to chromosome region 1q21. Proc Natl Acad Sci USA 86:4848–4852
Meek RL, Lonsdale-Eccles JD, Dale BA (1983) Epidermal filaggrin is synthesized on a large messenger ribonucleic acid as a high molecular weight precursor. Biochemistry 22:4867–4871
Mehrel T, Hohl D, Rothnagel JA, Longley MA, Bundman D, Cheng C, Lichti U, Bisher ME, Steven AC, Steinert PM, Yuspa SH, Roop DR (1990) Identification of a major keratinocyte cell envelope protein, loricrin. Cell 61:1103–1112
Ohno J, Fukuyama K, Hara A, Epstein WL (1989) Immuno- and enzyme-histochemical detection of phosphqprotein phosphatase in rat epidermis. J Histochem Cytochem 37:629–634
Peracchia C, Mittler BS (1972) Fixation by means of glutaraldehyde-hydrogen peroxide reaction products. J Cell Biol 53:234–238
Resing KA, Walsh KA, Dale BA (1984) Identification of two intermediates during processing of profilaggrin in neonatal mouse epidermis. J Cell Biol 99:1372–1378
Resing KA, Walsh KA, Haugen-Schofield J, Dale BA (1989) Identification of proteolytic cleavage sites in the conversion of profilaggrin in mammalian epidermis. J Biol Chem 264:1837–1845
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Rothnagel JA, Mehrel T, Idler WW, Roop DR, Steinert PM (1987) The gene for mouse epidermal filaggrin precursor. J Biol Chem 262:15643–15648
Scott IR, Harding CR (1986) Filaggrin breakdown to water binding compounds during development of the rat stratum corneum is controlled by the water activity of the environment. Dev Biol 115:84–92
Scott IR, Harding CR, Barrett JG (1982) Histidine-rich protein of the keratohyaline granules — source of the free amino acids, urocanic acid and pyrrolidone carboxylic acid in the stratum corneum. Biochim Biophys Acta 719:110–117
Steven AC, Bisher ME, Roop DR, Steinert PM (1989) Biosynthetic pathways of filaggrin and loricrin elucidated by immunolabelling of newborn mouse epidermis. J Cell Biol 109:258a
Sybert VP, Dale BA, Holbrook KA (1985) Ichthyosis vulgaris: identification of a defect in synthesis of filaggrin correlated with an absence of keratohyaline granules. J Invest Dermatol 84:191–194
Tezuka T, Takahashi M (1986) A novel protein of rat epidermis — haematoxylin stain-positive protein of keratohyaline granules as the membrane lining protein of the stratum corneum. J Dermatol (Tokyo) 13:417–425
Ugel AR (1969) Keratohyalin: extraction and in vitro aggregation. Science 166:250–251
Warhol MJ, Roth J, Lucocq JM, Pinkus GS, Rice RH (1985) Immuno-ultrastructural localization of involucrin in squamous epithelium and cultured keratinocytes. J Histochem Cytochem 33:141–149
Wells RS, Kerr CB (1965) Genetic classification of ichthyosis. Arch Dermatol 92:1–6
Williams ML (1983) The ichthyoses — pathogenesis and prenatal diagnosis: a review of recent advances. Pediatr Dermatol 1:1–24
Zettergren JG, Peterson LL, Wuepper KD (1984) Keratolinin: the soluble substrate of epidermal transglutaminase from human and bovine tissue. Proc Natl Acad Sci USA 81:238–242
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Günzel, S., Weidenthaler, B., Hau\er, I. et al. Keratohyalin granules are heterogeneous in ridged and non-ridged human skin: evidence from anti-filaggrin immunogold labelling of normal skin and skin of autosomal dominant ichthyosis vulgaris patients. Arch Dermatol Res 283, 421–432 (1991). https://doi.org/10.1007/BF00371777
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00371777