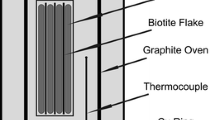
Abstract
Phase relations for the magnesio-hornblende bulk composition, 2 CaO·4 MgO·Al2O3·7 SiO2+ excess H2O, have been investigated to 10 kb employing hydrothermal and piston-cylinder techniques. The low-temperature limit of amphibole in this system lies at 519° C, 1,000 bars, 541° C, 2,000 bars, and 718° C, 10 kb. The low-T assemblage consists of an+chl+di+tc(+f), and is related to the adjacent high-T equilibrium assemblage, amph+an+chl+f, by the solid-solid reaction (A): 2 di+tc=tr. Small amounts of aluminum, hypothesized to be preferentially dissolved in the cpx (and in the tc) relative to amph, may account for the broad P-T stability range of the di+tc assemblage in the synthetic work relative to systems involving stoichiometric tr, ∘Ca2Mg5Si8O22(OH)2, such as are common in natural, Al-poor calc-silicate parageneses. Alternatively, the low-temperature assemblage produced in the experiments may be metastable. For the investigated bulk composition, synthetic tremolitic-cummingtonitic amphibole contains relatively modest amounts of ts, ∘Ca2Mg3Al2 IVSi6-Al2 IVO22(OH)2; at pressures of 1,000–3,000 bars, solid solution extends from near tremolite only to about cu11tr69ts20, analogous to most analyzed natural magnesio-hornblendic specimens. At 10 kb fluid pressure, the solid solution reaches approximately cu06tr53ts41 for the investigated bulk composition, and appears to be virtually independent of temperature. Amphibole and 14 Å chl react within the amphibole stability field, along curve (B), at about 704° C and 2,000 bars, to produce an, en, fo and f (ΔH=40.9 kcal/ mole); at pressures greater than approximately 7kb, due to the incompatibility of an and fo, the higher temperature assemblage consists of amph, an, en, sp and f. Above P fluid− T curve (B), the amphibole coexists with an+en+fo+f at low pressures; at higher pressures, the amphibole, which is in equilibrium with an+en+sp+f, is relatively more aluminous. The high-T stability limit of aluminous tr+fo lies approximately 20–25° C below the dehydration curve for stoichiometric tremolite on its own bulk composition. Reaction (C), tr+fo=2 di+5 en+f (ΔH = 39.4 kcal/mole), produces an+di+en+f, the highest temperature subsolidus assemblage investigated for the tr50ts50 bulk composition. Hydrous melt is encountered at temperatures at least as low as 900° C at 10 kb, and at that fluid pressure coexists with amphibole over an interval of more than 60° C. Limited solid solution observed between tr and ts in nature (tr100-70) is accounted for by the restricted range of amphibole compositions produced in the present study. Such amphiboles, moreover, appear to have both high- and low-temperature stability limits, as demonstrated by the experimental results.
Similar content being viewed by others
References
Boyd FR (1959) Hydrothermal investigations of amphiboles pp 377–396. In: Researches in geochemistry, Abelson PH (ed). Wiley, New York, 511p
Boyd FR, England JL (1960) Apparatus for phase-equilibrium measurements at pressures up to 50 kilobars and temperatures up to 1,750° C. J Geophys Res 65:741–748
Burnham CW, Holloway JR, Davis NF (1969) Thermodynamic properties of water to 1,000° C and 10,000 bars. Geol Soc Amer Special Paper 132:96 p
Cameron M, Papike JJ (1979) Amphibole crystal chemistry: a review. Fortschr Mineral 57:28–67
Colville P, Ernst WG, Gilbert MC (1966) Relationships between cell parameters and chemical compositions of monoclinic amphiboles. Am Mineral 51:1727–1754
Ernst WG (1968) Amphiboles: Springer, Berlin Heidelberg NewYork, 125 p
Fawcett JJ, Yoder HS (1966) Phase relationships of chlorites in the system MgO-Al2O3-SiO2-H2O. Am Mineral 51:351–380
Finnerty TA, Waychunas GA, Thomas WM (1978) The preparation of starting mixes for mineral syntheses by a freeze-dry technique. Am Mineral 63:415–418
Gilbert MC, Heiz RT, Popp RK, Spear FS (1982) Experimental studies of amphibole stability, p 229–353. In: Veblen DR, Ribbe PH (eds), Amphiboles: Petrology and experimental phase relations. Rev Mineral 9B:390 p
Graham CM, Navrotsky A (1986) Thermochemistry of the tremolite-edenite amphiboles with applications to amphibole-plagioclase-quartz equilibria. Contrib Mineral Petrol 93:18–32
Greenwood HJ (1979) Thermodynamic properties of edenite. Current Res B, Geol Survey Can 79-1B:365–370
Hallimond AF (1943) On the graphical representation of the calciferous amphiboles. Am Mineral 28:65–89
Hawthorne FC (1983) The crystal chemistry of the amphiboles. Can Mineral 21:173–480
Helgeson HC, Delany JM, Nesbitt HW, Bird DK (1978) Summary and critique of thermodynamic properties of rock-forming minerals. Am J Sci 278 A:229 p
Holloway JR (1973) The system pargasite-H2O-CO2: a model for melting of a hydrous mineral with a mixed-volatile fluid — I Experimental results to 8 kbar. Geochem Cosmochim Acta 36:651–666
Jasmund K, Schäfer R (1972) Experimentelle Bestimmung der P-T-Stabilitätsbereiche in der Mischkristallreihe Tremolit-Tschermakit. Contrib Mineral Petrol 34:101–115
Jenkins DM (1983) Stability and composition relations of calcic amphiboles in ultramafic rocks. Contrib Mineral Petrol 83:375–384
Jenkins DM (1984) Tschermakite: stable or metastable? Geol Soc Am Abstracts with Programs, 16:551
Kushiro I, Yoder HS Jr (1966) Anorthite-forsterite and anorthite-enstatite reactions and their bearing on the basalt-eclogite transformation. J Petrol 7:337–362
Leake BE (1968) A catalog of analyzed calciferous and subcalciferous amphiboles together with their nomenclature and associated minerals. Geol Soc Am Spec Paper 98:210 p
Leake BE (1978) Nomenclature of amphiboles. Can Mineral 16:501–520
Lindsley DH (1983) Pyroxene thermometry. Am Mineral 68:447–493
Misch P, Rice JM (1975) Miscibility of tremolite and hornblende in progressive Skagit metamorphic suite, North Cascades, Washington. J Petrol 16:1–21
Na KC, McCauley ML, Crisp JM, Ernst WG (1986) Phase relations to 3 kb in the system edenite+H2O and edenite+excess quartz+H2O. Lithos in press
Oba T (1978) Phase relationship of Ca2Mg3Al2Si6Al2O22(OH)2-Ca2Mg3Fe2 3+Si6Al2O22(OH)2 join at high temperature and high pressure — the stability of tschermakite. J Fac Sci Hokkaido Univ Ser IV, 18:339–350
Oba T (1980) Phase relations in the tremolite-pargasite join. Contrib Mineral Petrol 71:247–256
Obata M (1976) The solubility of Al2O3 in orthopyroxene in spinel and plagioclase peridotites and spinel pyroxenite. Am Mineral 61:804–816
O'Hara MJ (1967) Mineral parageneses in ultrabasic rocks, pp 393–403. In: Wyllie PJ (ed), Ultramafic and related rocks. Wiley, New York, 464 p
Orville PM, Greenwood HJ (1965) Determination of H of reaction from experimental pressure-temperature curves. Am J Sci 263:678–683
Papike JJ (ed) (1969) Pyroxenes and amphiboles: crystal chemistry and phase petrology. Mineral Soc Am Special Paper 2:314 p
Robie RA, Hemingway BS, Fisher JR (1978) Thermodynamic properties of minerals and related substances at 298.15 K and 1 bar (105 Pascals) pressure and at higher temperatures. US Geol Survey Bull 1452:456 p
Semet M, Ernst WG (1981) Experimental stability relations of the hornblende magnesiohastingsite. Geol Soc Am Bull 92:I, 71–74, II, 274–357
Sen G (1985) Experimental determination of pyroxene compositions in the system-CaO-MgO-Al2O3-SiO2 at 900–1,200° C and 10–15 kbar using PbO and H2O fluxes. Am Mineral 70:678–695
Skippen G, McKinstry BW (1985) Synthetic and natural tremolite in equilibrium with forsterite, enstatite, diopside and fluid. Contrib Mineral Petrol 89:256–262
Spear FS (1981) An experimental study of hornblende stability and compositional variability in amphibolite. Am J Sci 281:697–734
Sundius N (1946) The classification of the hornblendes and the solid solution relations in the amphibole group. Sver Geol Unders Arsb 40: no. 4, 1–36
Tagiri M (1973) Metamorphism of Paleozoic rocks in the Hitachi District, southern Abukuma Plateau, Japan. Sci Repts, Tohoku Univ, Ser III, 12:67 p
Thomas WN (1982) Stability relations of the amphibole hastingsite. Am J Sci 282:136–164
Turner FJ (1981) Metamorphic Petrology: Mineralogic, field and tectonic aspects. McGraw-Hill, New York, 524 p
Tuttle OF (1949) Two pressure vessels for silicate-water studies. Bull Geol Soc Amer 60:1727–1729
Veblen DR (ed) (1981) Amphiboles and other hydrous pyriboles-mineralogy. Rev Mineral 9A:372 p
Veblen DR, Ribbe PH (eds) (1982) Amphiboles: Petrology and experimental phase relations. Rev Mineral 9B:390 p
Welch MD, Graham CM, Trim H (1984) The reaction: tremolite+albite edenite+4 quartz, pp 140–141: In: Progress in experimental petrology. N.E.R.C.
Westrich HR, Holloway JR (1981) Experimental dehydration of pargasite and calculation of its entropy and Gibbs energy. Am J Sci 281:922–934
Author information
Authors and Affiliations
Additional information
Institute of Geophysics and Planetary Physics Publication No. 2811
Rights and permissions
About this article
Cite this article
Rong-Iong, C., Ross, C. & Ernst, W.G. Experimental studies to 10 kb of the bulk composition tremolite50-tschermakite50+excess H2O. Contr. Mineral. and Petrol. 93, 160–167 (1986). https://doi.org/10.1007/BF00371317
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00371317