Abstract
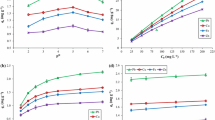
Biosorption technique was used for removal of cadmium under different conditions from water environment using a biosorbent, Volveriella volvaceas, locally growing fruit bodies of mushroom. Effects of different parameters like pH, sorbent concentration, ionic strength on the removal efficiency of cadmium by V. volvacea were carried out in continuation with adsorption kinetics and equilibrium isotherm experiments. From the kinetics studies it was found that nearly 95% of the total cadmium removal was achieved from cadmium spiked distilled water within first 15 minutes. Isotherm data was best fitted to linearised Langmuir equation and the sorption capacity was found to be varying from 9.13 to 9.33 mg/g for different sizes of sorbent. The uptake of cadmium(II) is a function of pH of the solution and increases with the increasing pH. Increasing ionic strength and the presence of soluble complexing agents such as ethylene diamine tetraacetic acid (EDTA) decrease the sorption of cadmium (II). The presence of other diavalent cations like calcium and magnesium impedes the uptake of cadmium (II). The presence of chloride ion has no significant effect on cadmium (II) removal. The spent biosorbent can effectively be regenerated with acid and can then be reused.
Similar content being viewed by others
References
Brierly, J.A.; Brierly, C.L.; Goyak, G.N.: AMT BIOCLAIM — A new waste water treatment and metal recovery technology. In Lawrence, R.M.; Branion, R.M.R.; Ebner, H.G. (eds.): Fundamental and applied biohydrometallurgy. pp. 291–303. Elsevier, Amsterdam, The Netherlands, 1987
Browing, E.M.D.: Toxicology of Industrial Metals. 2nd Ed. London: Butterworth Pub. 1969
Dey, S.; Rao, P.R.N.; Bhattacharya, B.C.; Bandyopadhyay, M.: Sorption of heavy metals by four Basidiomycetous fungi. Bioprocess Engg. 12 (1955) pp. 273–277
Emsley, J.: The Elements. Oxford: Clarendon Press. 1989
Helfferich, F.: Ion exchange. pp. 72–94. New York: McGraw Hill. 1962
Msaky, J.J.; Calvet, R.: Adsorption behavior of copper and zinc in soils: Influence of pH on adsorption characteristics. Soil Sci. 150(2) (1990) 513–522
Tobin, J.M.; Cooper, D.J.; Neufeld, R.J.: Uptake of metal ions by Rhizopus arrhizus biomass. Appl. Environ. Microbiol. 47. 4. (1984) 821–824
Townsley, C.C.; Ross, I.S.; Atkins, A.S.: Biorecovery of metallic residues from various industrial effluents using filamentous fungi. In Lawrence, R.M.; Branion, R.M.R.; Ebner, H.G. (eds.): Fundamental and applied biohydrometallurgy. Elsevier, Amsterdam, The Netherlands, 1987
Tsezos, M.; Volesky, B.: The mechanism of uranium biosorption by Rhizopus arrhizus. Biotechnol. Bioengg. 24 (1982) 385–401
Venkobachar, C.: Metal removal by waste biomass to upgrade wastewater treatment plants. Wat. Sci. Tech. 22 7/8 (1990) 319–320
Author information
Authors and Affiliations
Additional information
The present work was carried out by the financial support in terms of fellowship under the cultural exchange programme of the Indo-Bangladesh government. Special thanks to the Director, Bangladesh Institute of Technology, Dhaka, Bangladesh, for providing leave, which enabled the author in carrying out the research work.
Rights and permissions
About this article
Cite this article
Osman, M.S., Bandyopadhyay, M. Cadmium removal from water environment by a fungus Volveriella volvacea. Bioprocess Engineering 14, 249–254 (1996). https://doi.org/10.1007/BF00369921
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00369921