Abstract
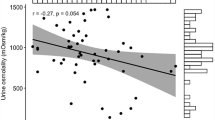
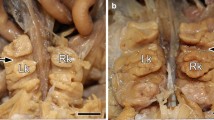
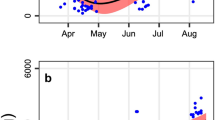
The white-tailed prairie dog is an obligate hibernator that enters a heterothermic phase when maintained in the cold with low intensity light and ad libitum food and water. The black-tailed prairie dog (a facultative hibernator) will not hibernate under similar conditions. It has been suggested that the black tailed prairie dog remains active during the winter because it can conserve water more effectively due to a more efficient kidney. The present study revealed no significant differences between the species in renal morphology: relative medullary thickness, nephron heterogeneity, renal vasculature, or fornix dimensions, all of which are structures associated with the urinary concentrating mechanism. In addition, there was no difference in number of nephrons between the two species. The black-tailed prairie dog does produce a more concentrated urine when food and water deprived. However, this difference was not observed when the animals were salt loaded. The water-deprivation and salt-loading experiments suggest that the higher urine osmolality produced by the back-tailed prairie dog during fasting is a result of a higher urea load due to a greater protein catabolism and not because of a differential capacity to concentrate urine.
Similar content being viewed by others
Abbreviations
- C :
-
cortex
- GFR :
-
glomerular filtration rate
- H :
-
height
- IS :
-
inner stripe
- IZ :
-
inner zone of medulla
- L :
-
length
- OS :
-
outer stripe
- PE :
-
polythylene
- RMT :
-
relative medullary thickness
- T a :
-
ambient temperature
- W :
-
width
References
Altschuler EM, Nagle RB, Braun EJ, Lindstedt SL, Krutzsh PH (1979) Morphological study of the desert heteromyoid kidney with emphasis on the genus Perognathus. Anat Rec 194: 461–468
Bakko EB (1977) Field water balance performance in prairie dogs (Cynomys leucurus and C. ludovicianus). Comp Biochem Physiol 56: 443–451
Bankir L, deRouffignac C (1985) Urinary concentrating ability: insights from comparative anatomy. Am J Physiol 249: R643-R666
Bargman J, Leonard SL, McNeely E, Robertson C, Jamison RL (1984) Examination of transepithelial exchange of water and solute in the rat renal pelvis. J Clin Invest 74: 1860–1870
Barrett JM, Kriz W, Kaissling B, deRouffignac C (1979) The ultrastructure of the nephrons of the desert rodent, Psammomys obesus, kidney. I. Thin limbs of Henle of short-looped nephrons. Am J Anat 151: 487–498
Battilana CA, Dobyan DC, Lacy FB, Bhattachara J, Johnston PA, Jamison RL (1978) Effect of chronic potassium loading on potassium secretion by the pars recta or descending limb of the juxtamedullary nephron in the rat. J Chin Invest 62: 1093–1103
Beuchat CA (1990) Body size, medullary thickness and urine concentrating ability in mammals. Am J Physiol 258: R298-R308
Berry CA (1982) Heterogeneity of tubular transport processes in the nephron. Ann Rev Physiol 44: 181–201
Bintz GL, Strand CE (1983) Nitrogen catabolism during starvation and starvation with water deprivation in Richardson's ground squirrel. J Comp Phsiol 149: 565–572
Bintz GL (1984) Water balance, water stress, and the evolution of seasonal torpor in ground-dwelling Sciurids. In: Murie JO, Michner GR (eds). The biology of ground dweling squirrels. University of Nebraska Press, Lincoln
Bonventre JV, Roman RJ, Lechene CP (1980) Effect of urea concentration of pelvic fluid on renal concentrating ability. Am J Physiol 239: F609-F618
Cahill GF, Owen OE (1967) Starvation and survival. Am Clim Ass Trans 79: 13–20
Calder WA (1984) Size, function and life history. Harvard University Press, Cambridge, Mass and London, England
Davis DE (1976) Hibernation and circannual rhythms of food consumption in marmots and ground squirrels. Q Rev Biol 51: 477–514
deRouffignac C (1990) Urinary concentrating mechanisms. In: Kinne RKH (ed) Comparative physiology, vol 2. Urinary concentrating mechanisms. Karger, Basel
Dewey GL, Elias H, Appel KP (1966) Stereology of the renal corpuscles of desert and swamp deer mice. Nephron 3: 352–365
Harlow HJ (1995) Fasting biochemistry of representative spontaneous and facultative hibernators: the white-tailed prairie dog and the black-tailed prairie dog. Physiol Zool (in press)
Harlow HJ, Menkens GE (1986) A comparison of hibernation in the black tailed prairie dog, white tailed prairie dog and Wyoming ground squirrel. Can J Zool 64: 793–796
Harlow HJ, Seal US (1982) Changes in hematology and metabolism in the serum and urine of the badger, Taxidea taxus, during food deprivation. Can J Zool 59: 2123–2128
Heisinger JF, King TS, Halling HW, Fields BL (1977) Renal adaptations to macro- and micro-habitats in the family Cricetidae. Comp Biochem Physiol 44: 767–774
Heisinger JF, Breitenbach RP (1969) Renal structural characteristics as indexes of renal adaptation for water conservation in the genus Sylvilagus. Physiol Zool 42: 160–172
Kaissling B, deRouffignac C, Barrett JM, Kriz W (1975) The structural organization of the kidney of the desert rodent, Psammonys obesus. Anat Embryol 148: 121–143
Moy RM (1971) Renal function in the hibernating ground squirrel Spermophilus columbianus. Am J Physiol 220: 747–753
Mrosovsky N (1978) Circannual cycles in hibernators. In: Wang CH, Hudson JW (ed) Strategies in cold: natural torpidity and thermogenesis. Academic Press, New York, pp 21–65
Munkacsi I, Palkovits M (1965) Volumetric analysis of glomerular size in kidneys of mammals living in desert, semidesert and water-rich environment in the Sudan. Circ Res 17: 303–311
Pizzimenti JJ (1975) Evolution of the prairie dog genus Cynomys. Occasional Papers of the Museum of Natural History, University of Kansas, Lawrence, Kansas 39: 1–73
Powell RA (1982) Prairie dog coloniality and black footed ferrets. Ecology 63: 1967–1968
Rickart EA (1989) Variation in renal structure and urine concentrating capacity among ground squirrels of the Spermophilus townsendii complex (Rodentia: Sciuridae). Comp Biochem Physiol 92A: 531–534
Rytand DA (1938) The number and size of mammalian glomeruli as related to kidney and to body weight, with methods for their enumeration and measurement. Am J Anat 62: 507–520
Schmidt-Nielsen B (1990) Function of the renal pelvis. In: Kinne RKH (ed) Urinary concentrating mechanisms, Comparative physiology, Vol 2. Karger, Basel pp 103–140
Schmidt-Nielsen B (1987) The renal pelvis. Kidney Int 31: 621–628
Schmidt-Nielsen B, O'Dell R (1961) Structure and concentrating mechanisms in the mammalian kidney. Am J Physiol 22: 1119–1124
Schmidt-Nielsen B, Churchill M, Reinking LN (1980) Occurrence of renal pelvic refluxes during rising urine flow rate in rats and hamsters. Kidney Int 18: 419–431
Schutz W, Schnermann J (1972) Pelvic urine concentration as a determinant of inner medullary solue concentration and urine osmolality. Pflügers Arch 334: 154–166
Sperber I (1994) Studies on the mammalian kidney. Bidrag fran Uppsala, 22: 249–431
Stewart J (1975) Urea handling by the renal countercurrent system: insights for computer simulation. Pflügers Arch 356: 133–151
Stewart J, Valtin H (1972) Computer simulation of osmotic gradient without active transport in renal inner medulla. Kidney Int 2: 264–270
Thompson TA, Agar MW, Bintz GL (1993) Lipid deposition and use by black-tailed prairie dogs, Cynomys ludovicianus in the natural environment. Physiol Zool 66: 561–579
Waugh WW (1977) Photometry of inulin and polyfructosan by use of a cystein/tryptophan reaction. Clin Chem 23: 639–645
Weisser F, Lacy FB, Weber H, Jamison RL (1970) Renal function in the chinchilla. Am J Physiol 219: 1706–1713
Author information
Authors and Affiliations
Additional information
Communicated by L.C.-H. Wang
Rights and permissions
About this article
Cite this article
Harlow, H.J., Braun, E.J. Kidney structure and function of obligate and facultative hibernators: the white-tailed prairie dog (Cynomys leucurus) and the black-tailed prairie dog (Cynomys ludovicianus). J Comp Physiol B 165, 320–328 (1995). https://doi.org/10.1007/BF00367316
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00367316