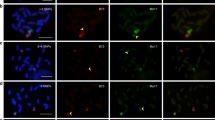
Abstract
The analysis of major satellite sequence differences between Mus spretus and laboratory mice provides a robust method for analyzing the centromere location for the genetic maps of each mouse chromosome. Fluorescence in situ hybridization (FISH) of a genomic probe, pMR196, for the laboratory mouse major satellite sequences was used to identify C57BL/6Ros (B6) pericentromeric heterochromatin in progeny of reciprocal backcross matings. These included 80 (B6xM. spretus)F1xM. spretus progeny (BSS) and 70 (B6xM. spretus)F1xB6 (BSB) progeny. FISH analysis of pericentromeric heterochromatin was conducted on the same metaphase spreads that were karyotypically analyzed for chromosomespecific banding patterns. Analysis of chromosomal segregation suggested that there was not primary deviation from random assortment during meiosis in the interspecific hybrid female, because nearly all of the 190 pair-wise comparisons did not deviate from expected and because there was no consistent pattern of deviation of the same chromosomes in the reciprocal backcross progeny from similar (C57BL/6xM. spretus)F1 hybrid females. These results affirm the value of using the major satellite to genetically mark pericentromeric heterochromatin in the analysis of the segregation and assortment of centromeres in Mus interspecific crosses.
Similar content being viewed by others
References
Brannan, C.I., Gilbert, D.J., Ceci, J.D., Matsuda, Y., Chapman, V.M., Mercer, J.A., Eisen, H., Johnston, L.A. Copeland, N.G., Jenkins, N.A. (1992). An interspecific linkage map of mouse chromosome 15 positioned with respect to the centromere. Genomics 13, 1075–1081.
Cattanach, B.M. (1978). Crossover suppression in mice heterozygous for tobacco mouse metacentrics. Cytogenet. Cell Genet. 20, 264–281.
Ceci, J.D., Matsuda, Y., Grubber, J.M., Jenkins, N.A., Copeland, N.G., Chapman, V.M. (1992). Interspecific backcrosses provide an important new tool for centromere mapping in mouse. In Sixth International Mouse Genome Conference, October 11–15, Buffalo, N.Y.
Ceci, J., Siracusa, L.D., jenkins, N.A., Copeland, N.G. (1990). A molecular genetic linkage map of mouse chromosome 4 including the localization of several protooncogenes. Genomics 5, 699–709.
Chapman, V.M., Kratzer, P.G., Quarantillo, B.A. (1983). Electrophoretic variation for X-chromosome-linked hypoxanthine phosphoribosyltransferase (HPRT) in wild-derived mice. Genetics 103, 785–795.
Chapman, V.M., Stephenson, D.A., Mullins, L.J., Keitz, B.T., Disteche, C., Orkin, S.H. (1991). Linkage of the erythroid transcription factor gene (Gf-1) to the proximal region of the X chromosome of mice. Genomics 9, 309–313.
Chapman, V.M., Hirotusne, S., Okazaki, Y., Hatada, I., Akasako, A., Mukai, T., Kawai, J., Hirasawa, T., Nishitani, Y., Watanabe, S., Shiroishi, T., Moriwaki, K., Matsuda, Y., Manly, K., Elliott, R., Hayashizaki, Y. (1992). Mouse genome mapping in an interspecific cross using restriction landmark genomic scanning (RLGS). In Sixth International Mouse Genome Conference, October 11–15, Buffalo, N.Y.
Cui, X., Gerwin, J., Navidi, W., Li, H., Kuehn, M., Arneim, N. (1992). Gene-centromere linkage mapping by PCR analysis of individual oocytes. Genomics 13, 713–717.
Davisson, M.T., Akeson, E.C. (1993). Recombination suppression by heterozygous Robertsonian chromosomes in the mouse. Genetics 133, 649–667.
Davisson, M.T., Roderick, T.H., Doolittle, D.P. (1989). Recombination percentages and chromosomal assignments. In Genetic Variants and Strains of the Laboratory Mouse, M.F. Lyon, A.G. Searle, eds (London: Oxford University Press), pp 432–505.
Eicher, E.M. (1978). Murine ovarian teratomas and parthenotes as cytogenetic tools. Cytogenet. Cell Genet. 20, 232–239.
Eppig, J.T., Eicher, E.M. (1983). Application of the ovarian teratoma mapping method in the mouse. Genetics 103, 797–812.
Eppig, J.T., Eicher, E.M. (1988). Analysis of recombination in the centromere region of mouse chromosome 7 using ovarian teratoma and backcross methods. J. Hered. 79, 425–429.
Hastie, N.D. (1989). Highly repeated DNA families in the genome of Mus musculus. In Genetic Variants and Strains of the Laboratory Mouse, M.F. Lyon, A.G. Searle, eds. (London: Oxford University Press), pp 559–573.
Hayashizaki, Y., Hirotusne, S., Hatada, I., Okazaki, Y., Komatsubara, H., Akasako, A., Shibata, I., Mukai, T., Kawai, J., Hirasawa, T., Nishitani, Y., Hirose, K., Watanabe, S., Manly, K., Elliott, R., Taylor, B., Chapman, V. (1992). Restriction Landmark Genomic Scanning (RLGS) and its application to mouse genome mapping with recombinant inbred strains. In Sixth International Mouse Genome Conference, October 11–15, Buffalo, N.Y.
Justice, M.J., Siracusa, L.D., Gilbert, D.J., Heisterkamp, N., Groffen, J., Chada, K., Silan, C.M., Copeland, N.G., Jenkins, N.A. (1990). A genetic linkage map of mouse chromosome 10: localization of eighteen molecular markers using a single interspecific backcross. Genetics 125, 855–866.
Justice, M.J., Gilbert, D.J., Kinzler, K.W., Vogelstein, B., Buchberg, A.M., Ceci, J.D., Matsuda, Y., Chapman, V.M., Patriotis, C., Makris, A., Tsichlis, P.N., Jenkins, N.A., Copeland, N.G. (1992). A molecular genetic linkage map of mouse chromosome 18 reveals extensive linkage conservation with human chromosomes 5 and 18. Genomics 13, 1281–1288.
Lawrence, J.B., Villnave, C.A., Singer, R.H. (1988). Sensitive, high-resolution chromatin and chromosome mapping in situ: presence and orientation of two closely integrated copies of EBV in a lymphoma line. Cell 52, 51–61.
Matsuda, Y., Chapman, V.M. (1991). In situ analysis of centromeric satellite DNA segregating in Mus species crosses. Mammalian Genome, 1, 71–77.
Matsuda, Y., Chapman, V.M. (1992). Analysis of sex-chromosome aneuploidy in interspecific backcross progeny between the laboratory mouse strain C57BL/6 and Mus spretus. Cytogenet. Cell Genet. 60, 74–78.
Matsuda, Y., Moens, P.B., Chapman, V.M. (1992). Deficiency of X and Y chromosomal pairing at meiotic prophase in spermatocytes of sterile interspecific hybrids between laboratory mice (Mus domesticus) and Mus spretus. Chromosoma 101, 483–492.
Pietras, D.F., Bennett, K.L., Siracusa, L.D., Woodworth-Gutai, M., Chapman, V.M., Gross, K.W., Kane-Haas, C., Hastie, N.D. (1983). Construction of a Mus musculus repetitive DNA library: identification of a new satellite sequence in Mus musculus. Nucleic Acids Res. 11, 6965–6983.
Ratty, A.K., Matsuda, Y., Elliott, R.W., Chapman, V.M., Gross, K.W. (1992). Genetic mapping of two DNA markers, D16Ros1 and D16Ros2, flanking the mutation site in the chakragati mouse, a transgenic insertional mutant. Mammalian Genome 3, 5–10.
Seldin, M.F., Howard, T.A., D'Eustachio, P. (1989). Comparison of linkage maps of mouse chromosome 12 derived from laboratory strain intraspecific and Mus spretus interspecific backcrosses. Genomics 5, 24–28.
Singer, M.F. (1982). Highly repeated sequences in mammalian genomes. Int. Rev. Cytol. 76, 67–112.
Siracusa, L.D., Alvord, W.G., Bickmore, W.A., Jenkins, N.A., Copeland, N.G. (1991). Interspecific backcross mice show sexspecific differences in allelic inheritance. Genetics 128, 813–821.
Sokal, R.R., Rohlf, F.J. (1981). Biometry, 2nd ed. (New York: W.H. Freeman and Company).
Takahashi, E, Hori, T., Lawrence, J.B., McNeil, J., Singer, R.H., O'Connell, P., Leppert, M., White, L. (1989). Human type II collagen gene (COL2A1) assigned to chromosome 12q13.1→q13.2 by in situ hybridization with biotinylated DNA probe. Jpn. J. Hum. Genet. 34, 307–311.
Takahashi, E., Hori, T., Sutherland, G.R. (1990). Mapping of the human type II collagen gene (COL2A1) proximal to fra(12)(q13.1) by nonisotopic in situ hybridization. Cytogenet. Cell Genet. 54, 84–85.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Matsuda, Y., Manly, K.F. & Chapman, V.M. In situ analysis of centromere segregation in C57BL/6 x Mus spretus interspecific backcrosses. Mammalian Genome 4, 475–480 (1993). https://doi.org/10.1007/BF00364780
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00364780