Summary
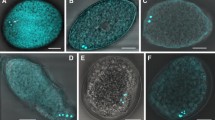
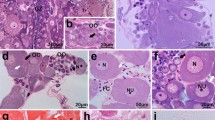
Tadpole development is eliminated in the life cycle of the ascidian Molgula pacifica. The elimination of a tailed larva is termed anural development, in contrast to urodele development which is exhibited by most ascidian species. In the present study, transmission electron microscopy and histochemistry were used to gain a better understanding of anural development in M. pacifica. The fine structure of M. pacifica oocytes and fertilized eggs was similar to urodele oocytes and eggs, except that a perivitelline space and test cells were absent. M. pacifica embryos exhibited the typical cleavage pattern of urodele embryos. Gastrulation was initiated at the vegetal pole, as in urodeles, and occurred at the same time as in two urodele species (Molgula manhattensis and Pyura haustor). However, changes in cell shapes and cell movements of the vegetal pole cells that participate in gastrulation were highly modified compared to commonly studied ascidians. The changes in shapes and movements of the vegetal pole cells were minimal and resulted in embryos having a very small archenteron and blastopore. The presence of large, yolky cells in the interior of the embryo likely restricted vegetal cell movements. Two ultrastructurally distinct types of epidermal cells were evident at the gastrula stage. When gastrulae were manually dechorionated from their surrounding mucous-follicular envelope layers, the embryos were already surrounded by a thin tunic. When day 1 juveniles in the process of hatching were sectioned along the anterior-posterior axis, regional differences in cell types were evident. Differentiated muscle cells in the posterior region were not evident. Day 1 M. pacifica juveniles, anural-developing M. provisionalis juveniles and tadpoles from three urodele species were tested for their abilities to express AchE activity. The highest levels of AchE activity were detected in the larval tail muscle cells of urodeles, low levels of activity were detected in the posterior region of M. provisionalis juveniles, whereas M. pacifica juveniles did not exhibit AchE activity. The results are discussed in terms of evolutionary mechanisms responsible for anural development in ascidians.
Similar content being viewed by others
References
Bates WR (1980) A scanning electron microscope study of the structure and fertilization of an ascidian egg, Ciona intestinalis. Can J Zool 58:1942–1946
Bates WR, Jeffery WR (1987) Localization of axial determinants in the vegetal pole of ascidian eggs. Dev Biol 124:65–76
Bates WR, Mallett JE (1991) Anural development of the ascidian Molgula pacifica (Huntsman). Can J Zool 69:618–627
Berrill NJ (1931) Studies in tunicate development. Part II. Abbreviation of Development in the Molgulidae. Phil Trans R Soc B 219:281–346
Berrill NJ (1935) Studies in tunicate development. Part III. Differential retardation and acceleration. Phil Trans R Soc B 225:255–326
Conklin EG (1905) The organization and cell lineage of the ascidian egg. J Acad Natl Sci 13:1–119
DeBeer GR (1940) Embryos and ancestors. Clarendon Press, Oxford
Damas D (1902) Recherces sur le developpment des Molgules. Arch Biol 18:599–664
Garstang WJ (1922) The theory of recapitulation: a critical restatement of the biogenic law. Zool J Linn Soc 35:81–101
Huntsman AG (1912) Ascidians from the coasts of Canada. Trans Can Instit 9:111–148
Karnovsky MJ, Roots L (1964) A ‘direct-coloring’ thiocholine method for cholinesterase. J Histochem. Cytochem 12:219–221
Kessel RG, Kemp NE (1962) An electron microscopy study on the oocyte, test cells and follicular envelope of the tunicate, Molgula manhattensis. Ultrastruct Res 6:57–76
Lacaze-Duthiers FJH de (1874) Histoire des ascidies simples des totes de France I. Arch Zool Exp Gen 3:119–656
Jeffery WR, Meier S (1983) A yellow crescent cytoskeletal domain in ascidian eggs and its role in early development. Dev Biol 96:125–143
Jeffery WR, Swalla BJ (1990) Anural development in ascidians: evolutionary modification and elimination of the tadpole larva. Sem Dev Biol 1:253–261
Jeffery WR, Swalla BJ (1991) An evolutionary change in the muscle lineage of an anural ascidian embryo is restored by interspecific hybridization with a urodele ascidian. Dev Biol 145:328–337
Millar RH (1954) The breeding and development of the ascidian Pelonaia corrugate Forbes and Goodsir. J Mar Biol Assoc 33:681–687
Millar RH (1962) The breeding and development of the ascidian Polycarpa tinctor. Q J Microsc Sci 103:399–312
Raff RA, Kaufmann TC (1983) Embryos, genes and evolution. Macmillan Press, New York, pp 1–395
Sawada T, Schatten G (1989) Effects of cytoskeletal inhibitors on ooplasmic segregation and microtubule organization during fertilization and early development in the ascidian Molgula occidentalis. Dev Biol 132:331–342
Satoh N, Deno T, Nishida H, Nishikata T, Makabe W (1990) Cellular and molecular mechanisms of muscle cell differentiation in ascidian embryos. Int Rev Cytology 122:221–258
Swalla BJ, Jeffery WR (1990) Interspecific hybridization between an anural and urodele ascidian: Differential expression of urodele features suggest multiple mechanisms control anural development. Dev Biol 142:319–334
Venuti JM, Jeffery WR (1989) Cell lineage and determination of cell fate in ascidian embryos. Int J Dev Biol 33:197–212
West AB, Lambert CC (1976) Control of spawning in the tunicate Styela plicata by variations in a natural light regime. J Exp Zool 195:263–270
Whittaker JR (1973) Segregation during ascidian embryogenesis of egg cytoplasmic information for tissue specific enzyme development. Proc Natl Acad Sci USA 70:2096–2100
Whittaker JR (1979) Development of vestigial tail muscle acetylcholinesterase in embryos of an anural ascidian species. Biol Bull 156:393–407
Whittaker JR (1987) Cell lineages and determinants of cell fate in development. Am Zool 27:607–622
Young CM, Gowan RE, Dalby J, Pennachetti CA, Gagliardi D (1988) Distributional consequences of adhesive eggs and anural development in the ascidian Molgula pacifica (Huntsman 1912). Biol Bull 174:39–46
Author information
Authors and Affiliations
Additional information
Offprint requests to: W.R. Bates
Rights and permissions
About this article
Cite this article
Bates, W.R., Mallett, J.E. Ultrastructural and histochemical study of anural development in the ascidian Molgula pacifica (Huntsman). Roux's Arch Dev Biol 200, 193–201 (1991). https://doi.org/10.1007/BF00361337
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00361337