Abstract
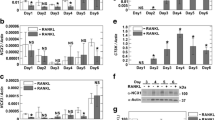
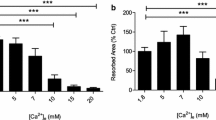
Carbonic anhydrase II (CA II), an enzyme catalyzing the interconversion of CO2 and water to HCO −3 and protons, has a key role in osteoclastic bone resorption, but little is known of the regulation of CA II gene expression by calcitonin. Analysis of mRNA in osteoclasts has been difficult because of the problems of obtaining sufficient number of purified osteoclasts from bone. In this study, however, we have investigated the regulation of CA II mRNA in rat osteoclasts and their putative mononuclear precursors by using in situ hybridization. We have found that the CA II gene is expressed at high levels in osteoclasts and what are probably their maturing mononuclear precursors. Measurement of CA II mRNA in cultured osteoclasts and their putative mononuclear precursor cells by cytophotometry provided evidence that calcitonin, a direct inhibitor of mammalian osteoclast activity, reduces the levels of CA II mRNA in a dose dependent manner; maximum reduction was observed at a concentration of 100pM of calcitonin. In addition, calcitonin reduced the number of CA II mRNA-positive mononuclear precursor cells. The results also suggest that expression of the CA II gene is a feature of cells committed to the osteoclast lineage.
Similar content being viewed by others
Abbreviations
- CA II :
-
Carbonic anhydrase II
- TRAcP :
-
tartrate-resistant acid phosphatase
- AcP :
-
acid phosphatase
- MOPC :
-
mononuclear osteoclast precursor cells
References
Arnett TR, Dempster DW (1987) A comparative study of disaggregated chicken and rat osteoclasts in vitro: effect of calcitonin and prostaglandins. Endocrinology 120: 602–608
Baron R (1989) Molecular mechanisms of bone resorption by osteoclasts. Anat Rec 224: 317–324
Baron R, Neff L, Tran Van P, Nefussi JR, Vignery A (1986) Kinetic and cytochemical identification of osteoclast precusors and their differentiation into multinucleated osteoclasts. Am J Pathol 122: 363–378
Billecocq A, Emanuel JR, Levenson R, Baron R (1990) 1α, 25-Dihydroxyvitamin D3 regulated the expression of carbonic anhydrase II in nonerythoid avaian bone marrow cells. Proc Natl Acad Sci USA 87: 6470–6474
Blair HC, Teitlebaum SL, Ghiselli R, Glucks (1989) Osteoclastic bone resorption by a polarized vacuolar proton pump. Science 245: 855–857
Brown D, Roth J, Kumpulainen T, Orci L (1982) Ultrastructural immunocytochemical localization of carbonic anhydrase. Histochemistry 75: 209–213
Cathala G, Savouret JF, Mendez B, West BL, Karin M, Martial JA, Baxter JD (1983) A method for isolation of intact, translationally active ribonucleic acid. DNA Cell Biol 2: 329–335
Chambers TJ, Revell PA, Fuller K, Athanasou NA (1984) Resorption of bone by isolated rabbit osteoclasts. J Cell Sci 66: 383–399
Everts V, Delaisse JM, Korper W, Niehof A, Vaes G, Beertsen W (1992) Degradation of collagen in the bone-resorbing compartment underlying the osteoclast involve both cysteine-proteinases and matrix metalloproteinases. J Cell Physiol 150: 221–231
Gay CV, Schraer H, Anderson RE, Cao H (1984) Current studies on the location and function of carbonic anhydrase in osteoclasts. Ann NY Acad Sci 429: 473–478
Gluck KS, Roodman GD (1990) Sequential expression of phenotype markers for osteoclasts during differentiation of precursors for multinucleated cells formed in long term human marrow culture. Endocrinology 127: 3215–3221
Hall TJ, Chambers TJ (1990) The Na+/H+ antiporter is the primary proton transport system used by osteoclasts during bone resorption. J Cell Physiol 142: 420–424
Hall TJ, Higgins W, Tardif C, Chambers TJ (1991) A comparison of the effect of inhibitors of carbonic anhydrase on osteoclastic bone resorption and purified carbonic anhydrase isoenzyme II. Calcif Tissue Int 49: 328–332
Marino LR, Muglia BH, Yamada T (1990) H+−K+-ATPase and carbonic anhydrase II gene expression in the developing rat fundus. Am J Physiol 258: G108-G115
Muallem SD, Blissard EJ, Cragoo E, Sachs G (1988) Activation of the Na+/H+ and Cl−/HCO −3 exchange by stimulation of acid secretion in the parietal cell. J Biol Chem 263: 14703–14711
Nicholson GC, Moseley JM, Serton FA, Mendelsohn PAD, Martin TJ (1986) Abundant calcitonin receptors in isolated rat osteoclasts: biochemical and autoradiographic characterization. J Clin Invest 78: 355–360
Nicholson GC, Moseley JM, Yate AFP, Martin TJ (1987) Control of cyclic adenosine 3′-5′-monophosphate production in osteoclasts: calcitonin-induced persistent activation and homologous desensitization of adenylate cyclase. Endocrinology 120: 1902–1908
Overgaard K, Riis BJ, Christiansen C (1989) Nasal calcitonin for treatment of established osteoporosis. Clin Endocrinol (Oxf) 30: 435–442
Prallet B, Male P, Neff L, Baron R (1992) Identification of a functional mononuclear precusor of the osteoclast in chicken medullary bone marrow cultures. J Bone Miner Res 7: 405–414
Raisz LC, Simmons HA, Thompson WJ, Shepard KL, Anderson PS, Rodan GA (1988) Effect of a potent carbonic anhydrase inhibitor on bone resorption in organ culture. Endocrinology 122: 1083–1086
Ries WL (1984) Osteogenic periosteum esterase activity: a comparative morphological and cytochemical study of bone cells in situ on rat proximal tibiae and in smears. J Histochem Cytochem 32: 55–62
Roth DE, Venta PJ, Tashian RE, Sly WS (1992) Molecular basis of human carbonic anhydrase II deficiency. Proc Natl Acad Sci USA 89: 1804–1808
Scheven BAA, Kawilarang-de Haas EWM, Wassenaar AM, Nijweide PJ (1986) Differentiation kinetics of osteoclasts in the periosteum of embryonic bones in vivo and in vitro. Anat Rec 214: 418–423
Sundquist KT, Leppilampi M, Jarvelin K, Kumpulainen T, Vaananen HK (1987) Carbonic anhydrase isoenzyme in isolated rat peripheral monocytes, tissues macrophages and osteoclasts. Bone 8: 33–38
Tashian RE, Hewett-Emmett D (1984) Biology and chemistry of the carbonic anhydrase. Ann NY Acad Sci 429: 1–64
Vaananen HK (1984) Immunohistochemical localization of carbonic anhydrase isoenzymes I and II in human bone, cartilage and giant cell tumour. Histochemistry 81: 485–487
Vaes G (1988) Cellular biology and biochemical mechanism of bone resorption. Clin Orthop 231: 239–271
Zaidi M, Datta HK, Moonga BS, Maclntyre I (1990) Evidence that the action of calcitonin on rat osteoclasts is mediated by G proteins acting via separate post-receptor pathways. J Endocrinol 126: 473–481
Zheng MH, Papadimitriou JM, Nicholson GC (1991) A quantitative cytochemical investigation of osteoclasts and multinucleate giant cells. Histochem J 23: 180–188
Zheng MH, Papadimitriou JM, Nicholson GC (1991a) RNA synthesis in isolated rat ostoeclasts: inhibitory effect of calcitonin. Bone 12: 317–322
Zheng MH, Nicholson GC, Warton A, Papadimitriou JM (1991b) What is new in osteoclast ontogeny? Pathol Res Pract 187: 117–125
Zheng MH, Wood DJ, Papadimitriou JM, Nicholson GC (1992) Evidence that protein kinase-A, calcium-calmodulin kinase and cytoskeletal proteins are involved in osteoclast retraction induced by calcitonin. Exp Mol Pathol 57: 105–115
Zheng MH, Fan Y, Wysocki S, Wood DJ, Papadimitriou JM (1993) Detection of mRNA for carbonic anhydrase II in human osteoclast-like cells by in-situ hybridization. J Bone Miner Res. 8: 111–116
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zheng, M.H., Fan, Y., Wysocki, S. et al. Carbonic anhydrase II gene transcript in cultured osteoclasts from neonatal rats: effect of calcitonin. Cell Tissue Res 276, 7–13 (1994). https://doi.org/10.1007/BF00354778
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00354778