Abstract
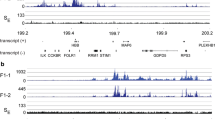
In vitro DNAase I footprinting and gel mobility shift assays have shown that the activities of several nuclear factors (GATA-1, Sp1) that bind to the promoter and downstream enhancer regions of the chicken histone H5 gene are reduced in mature erythrocytes relative to those in immature erythrocytes. In this study we investigated site occupancy in the promoter and downstream enhancer regions of the H5 gene in mature and immature erythrocytes. The ligation-mediated polymerase chain reaction was used to detect DNAase I footprints generated in situ. Most of the sites that bound to Sp1 and/or Sp1-like proteins and GATA-1 in the promoter and enhancer were occupied in situ in mature and immature erythrocytes. However, the level of protection at Sp1/Sp1-like binding sites in the H5 enhancer region of mature erythroid cells was generally less than that observed for immature cells, suggesting that for any given mature cell not all of the Sp1/Sp1-like binding sites are occupied. Nevertheless, the results of this study suggest that the enhancer and promoter of the H5 gene in mature erythrocytes should be functional, agreeing with nuclear runon studies showing transcriptional activity of the H5 gene in mature permeabilized cells.
Similar content being viewed by others
References
AffolterM, CôtéJ, RenaudJ, Ruiz-CarrilloA (1987) Regulation of histone and βA-globin gene expression during differentiation of chicken erythroid cells. Mol Cell Biol 7:3663–3672
CombM, GoodmanHM (1990) CpG methylation inhibits proenkephalin gene expression and binding of the transcription factor AP-2. Nucleic Acids Res 18:3975–3982
DavieJR, HendzelMJ (1994) Multiple functions of dynamic acetylation. J Cell Biochem 55:98–105
DelcuveGP, DavieJR (1989) Chromatin structure of erythroid-specific genes of immature and mature chicken erythrocytes. Biochem J 263:179–186
FaisstS, MeyerS (1992) Compilation of vertebrate-encoded transcription factors. Nucleic Acids Res 20:3–26
FengWC, SouthwoodCM, BiekerJJ (1994) Analyses of β-thalassemia mutant DNA interactions with erythroid Krüppel-like factor (EKLF), an erythroid cell-specific transcription factor. J Biol Chem 269:1493–1500
GallardaJL, FoleyKP, YangZ, EngelJD (1989) The β-globin stage selector element factor is erythroid-specific promoter/enhancer binding protein NF-E4. Genes Dev 3:1845–1859
GariglioP, BellardM, ChambonP (1981) Clustering of RNA polymerase B molecules in the 5′ moiety of the adult β-globin gene of hen erythrocytes. Nucleic Acids Res 9:2589–2598
GarrityPA, WoldBJ (1992) Effects of different DNA polymerases in ligation-mediated PCR: enhanced genomic sequencing and in vivo footprinting. Proc Natl Acad Sci USA 89:1021–1025
Gomez CuadradoA, RousseauS, RenaudJ, Ruiz CarrilloA (1992) Repression of the H5 histone gene by a factor from erythrocytes that binds to the region of transcription initiation. EMBO J 11:1857–1866
MerikaM, OrkinSH (1995) Functional synergy and physical interactions of the erythroid transcription factor GATA-1 with the Krüppel family proteins Sp1 and EKLF. Mol Cell Biol 15:2437–2447
MillerIJ, BiekerJJ (1993) A novel, erythroid cell-specific murine transcription factor that binds to the CACCC element and is related to the Krüppel family of nuclear proteins. Mol Cell Biol 13:2776–2786
MinieME, KimuraT, FelsenfeldG (1992) The developmental switch in embryonic rho-globin expression is correlated with erythroid lineage-specific differences in transcription factor levels. Development 115:1149–1164
MuellerPR, GarrityPA, WoldB (1994) Ligation-mediated PCR for genomic sequencing and footprinting. In: AusubelFM, BrentR, KingstonRE, MooreDD, SeidmanJG, SmithJA, StruhlK (eds) Current protocols in molecular biology 15 John Wiley & Sons, Seacaucus, New Jersey, pp 5.1–15.5.26
NeelinJM, CallahanPX, LambDC, MurrayK (1964) The histones of chicken erythrocyte nuclei. Can J Biochem 42:1743–1752
OmichinskiJG, CloreGM, SchaadO, FelsenfeldG, TrainorC, AppellaE, StahlSJ, GronenbornAM (1993) NMR structure of a specific DNA complex of Zn-containing DNA binding domain of GATA-1. Science 261:438–446
PennerCG, DavieJR (1994) Transcription factor GATA-1-multiprotein complexes and chicken erythroid development. FEBS Lett 342:273–277
PuruckerM, BodineD, LinH, McDonaghK, NienhuisAW (1990) Structure and function of the enhancer 3′ to the human Agamma globin gene. Nucleic Acids Res 18:7407–7415
RenaudJ, Ruiz CarilloA (1986) Fine analysis of the active H5 gene chromatin of chicken erythroid cells at different stage of differentiation. J Mol Biol 189:217–226
RidsdaleJA, DavieJR (1987) Chicken erythrocyte polynucleosomes which are soluble at physiological ionic strength and contain linker histones are highly enriched in β-globin gene sequences. Nucleic Acids Res 15:1081–1096
RidsdaleJA, RattnerJB, DavieJR (1988) Erythroid-specific gene chromatin has an altered association with linker histones. Nucleic Acids Res 16:5915–5926
RousseauS, AsselinM, RenaudJ, Ruiz-CarrilloA (1993) Transcription of the histone H5 gene is regulated by three differentiation-specific enhancers. Mol Cell Biol 13:4904–4917
SunJM, WiaderkiewiczR, Ruiz CarilloA (1989) Histone H5 in the control of DNA synthesis and cell proliferation. Science 245:68–71
SunJ-M, PennerCG, DavieJR (1992) Analysis of erythroid nuclear proteins binding to the promoter and enhancer elements of the chicken histone H5 gene. Nucleic Acids Res 20:6385–6392
SunJ-M, PennerCG, DavieJR (1993) Repression of histone H5 gene expression in chicken mature erythrocytes is correlated with reduced DNA-binding activities of transcription factors Sp1 and GATA-1. FEBS Lett 331:141–144
TrainorCD, StamlerSJ, EngelJD (1987) Erythroid-specific transcription of the chicken histone H5 gene is directed by a 3′ enhancer. Nature 328:827–830
Author information
Authors and Affiliations
Additional information
The first two authors have contributed equally to this work
Rights and permissions
About this article
Cite this article
Sun, JM., Ferraiuolo, R. & Davie, J.R. In situ footprinting of chicken histone H5 gene in mature and immature erythrocytes reveals common factor-binding sites. Chromosoma 104, 504–510 (1996). https://doi.org/10.1007/BF00352114
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00352114