Summary
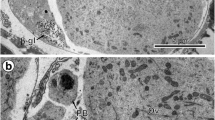
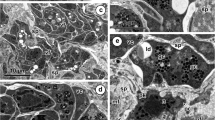
The origin of proteinaceous yolk in oocytes of Ciona, intestinalis appears to involve the activity of two kinds of vesicles derived from the Golgi complex. One kind of vesicle contains a granular product of considerable density while the contents of the other type of vesicle are of low density. Both types of vesicles become widely dispersed in the ooplasm during vitellogenesis. The high-density vesicle exhibits greater size variation than the lowdensity vesicle. The growing yolk globules possess an external often folded membrane enclosing both granular and vesicular elements. The granular-vesicular bodies are observed in wide size ranges and they appear to arise and increase in size by fusion or incorporation of numerous high-density vesicles, low-density vesicles, and smaller granular-vesicular bodies. The relationship of the developing yolk globules to ribosomes, pinocytotic vesicles, and vesicular endoplasmic reticulum is illustrated.
Similar content being viewed by others
Literature
Afzelius, B. A.: Electron microscopy of Golgi elements in sea urchin eggs. Exp. Cell Res. 11, 67–85 (1956).
Anderson, E.: Oocyte differentiation and vitellogenesis in the roach, Periplaneta americana. J. Cell Biol. 20, 131–155 (1964).
Balinsky, B. I., and R. J. Devis: Origin and differentiation of cytoplasmic structures in the oocytes of Xenopus laevis. Acta Embryol. Morph. exp. (Palermo) 6, 55–108 (1963).
Beams, H. W.: Cellular membranes in oogenesis. In: Cellular membranes in development, p. 175–219. New York: Academic Press 1964.
—, and R. G. Kessel: Intracisternal granules of the endoplasmic reticulum in the crayfish oocyte. J. Cell Biol. 13, 158–162 (1962).
—: Electron microscope studies on developing crayfish oocytes with special reference to the origin of yolk. J. Cell Biol. 18, 621–649 (1963).
Caro, L. G., and G. E. Palade: Protein synthesis, storage, and discharge in the pancreatic exocrine cell. An autoradiographic study. J. Cell Biol. 20, 473–495 (1964).
Davenport, R., and J. C. Davenbort: A cytochemical study of cytoplasmic basic proteins in the ascidian oocyte. J. Cell Biol. 25, 319–326 (1965).
Droller, M. J., and T. F. Roth: The endoplasmic reticulum, Golgi system, and micropinocytotic contributions to yolk formation in the guppy oocyte. J. Cell Biol. 23, 108 A (1964).
Favard, P., et N. Carasso: Origine et ultrastructure des plaquettes vitellines de la Planorbe. Arch. Anat. micr. Morph. exp. 47, 211–234 (1958).
Favard-Séréno, C.: Phenomène de pinocytose au cours de la vittellogenèse protéique chez le grillon (Orthoptère). J. Microscopie 3, 323–338 (1964).
Hope, J. H., A. A. Humphries, and G. H. Bourne: Ultrastructural studies on developing oocytes of the salamander Triturus viridescens. II. The formation of yolk. J. Ultrastruct. Res. 10, 547–556 (1964).
Hsu, W. S.: An electron microscopic study on the origin of yolk in the oocytes of the ascidian Boltenia villosa Stimpson. Cellule 62, 145–163 (1962).
Jamieson, J. D., and G. E. Palade: Intracellular transport of newly synthesized proteins in the exocrine pancreas. J. Cell Biol. 27, 47 A (1965).
Kessel, R. G.: The role of the Golgi complex in the formation of proteinaceous yolk in oocytes of the tunicates, Ciona and Styela. J. Cell Biol. 23, 119 A (1964).
—: Cytodifferentiation in ascidian oocytes. Anat. Rec. 151, 371 (1965).
—, and H. W. Beams: Micropinocytosis and yolk formation in oocytes of the small milkweed bug. Exp. Cell Res. 30, 440–443 (1963a).
- - Electron microscope studies on developing oocytes of the lobster, Homarus, with special reference to the method of yolk formation. J. Cell Biol. 19, 87 A (1963b).
Lanzavecchia, G.: The formation of yolk in frog oocytes. Proc. European Regional Conf. Elect. Micros., Delft 2, 746–749 (1960).
Locke, M., and J. V. Collins: The structure and formation of protein granules in the fat body of an insect. J. Cell Biol. 26, 857–884 (1965).
Luft, J. H.: Improvements in epoxy resin embedding methods. J. biophys. biochem. Cytol. 9, 409–414 (1961).
MacBride, E. W., and H. R. Hewer: Zoology. In: Recent advances in microscopy (A. Piney, Ed.), p. 88–167. Philadelphia: P. Blakiston Son & Co. 1931.
Mancuso, V.: Ultrastructural changes in the cytoplasm of Ciona intestinalis oocytes. Acta Embryol. Morph. exp. (Palermo) 7, 269–295 (1964).
Mazia, D., P. A. Brewer, and M. Alfert: The cytochemical staining and measurement of protein with mercuric bromphenol blue. Biol. Bull. 104, 57–67 (1953).
Millonig, G.: Advantages of a phosphate buffer for OsO4 solutions in fixation. J. appl. Phys. 32, 1637–1638 (1961).
Palade, G.: A study of fixation for electron microscopy. J. exp. Med. 95, 285 (1952).
Porter, K. R.: The ground substance; observations from electron microscopy. In: The cell (J. Brachet and A. E. Mirsky, Eds.), vol. 2, p. 621–675. New York: Academic Press 1961.
Raven, Ch. P.: Oogenesis: The storage of developmental information. New York: Pergamon Press 1961.
Reynolds, E. S.: The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 17, 208–211 (1963).
Roth, T. F., and K. R. Porter: Specialized sites on the cell surface for protein uptake. Proc. Intern Congr. Elect. Microsc., Philad., vol. 2, p. 114–115. New York: Academic Press 1962.
—: Yolk protein uptake in the oocyte of the mosquito, Aëdes aegypti. J. Cell Biol. 20, 313–332 (1964).
Sabatini, D. D., K. Bensch, and R. J. Barnett: Cytochemistry and electron microscopy. The preservation of cellular ultrastructure and enzymatic activity by aldehyde fixation. J. Cell Biol. 17, 19–58 (1963).
Siekevitz, P., and G. E. Palade: Cytochemical study on the pancreas of the guinea pig. I. Isolation and enzymatic activities of cell fractions. J. biophys. biochem. Cytol. 4, 203–218 (1958a).
—: Cytochemical study on the pancreas of the guinea pig. II. Functional variation in enzymatic activity. J. biophys. biochem. Cytol. 4, 309–318 (1958b).
—: Cytochemical studies on the pancreas of the guinea pig. VI. Release of enzymes and ribonucleic acid from ribonucleoprotein particles. J. biophys. biochem. Cytol. 7, 631–644 (1960).
Telfer, W. H.: The route of entry and localization of blood proteins in the oocytes of saturnid moths. J. biophys. biochem. Cytol. 9, 747–759 (1961).
Ward, R. T.: The origin of protein and fatty yolk in Rana pipiens. II. Electron microscopical and Cytochemical observations of young and mature oocytes. J. Cell Biol. 14, 309–341 (1962).
- Dual mechanisms for the formation of yolk platelets in Rana pipiens. J. Cell Biol. 23, 100 A (1964).
Wartenberg, H.: Experimentelle Untersuchungen über die Stoffaufnahme durch Pinocytose während der Vitellogenese des Amphibienoocyten. Z. Zellforsch. 63, 1004–1019 (1964).
Watson, M. L.: Staining of tissue sections for electron microscopy with heavy metals. J. biophys. biochem. Cytol. 4, 475–478 (1958).
Worley, L. G., and L. G. Moriber: The origin of protein yolk from the Golgi apparatus in gastropods. Trans. N. Y. Acad. Sci. 23, 352–356 (1961).
Yasuzumi, G., and H. Tanaka: Electron microscope studies on the fine structure of the ovary. I. Studies on the origin of yolk. Exp. Cell Res. 12, 681–685 (1957).
Author information
Authors and Affiliations
Additional information
This investigation was supported by research grants (GM-09229, HD-00699) from the National Instituts of Health, U. S. Public Health Service and a Career Development Award (GM-11,524) from the National Institute of General Medical Science.
Rights and permissions
About this article
Cite this article
Kessel, R.G. Electron microscope studies on the origin and maturation of yolk in oocytes of the tunicate, Ciona intestinalis . Zeitschrift für Zellforschung 71, 525–544 (1966). https://doi.org/10.1007/BF00349612
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00349612