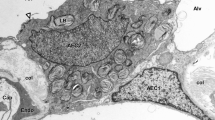
Summary
The formation of gap junctions was studied in pancreatic exocrine cells of rats and mice during late embryonic and neonatal development by the freeze-fracture replica method. Small gap junctions were present in association with tight junctional strands near the cell apex during embryonic development. Independently of tight junctions, small gap junctions were sometimes seen more basally on day 13 to 15 of gestation. The gap junctions increased in number and were rapidly enlarged by day 18 to 20 of gestation. Large particles 12–13 nm in diameter were frequently associated with the gap junction, which consisted of 10 nm particles. The large particles were either irregularly distributed or arranged in hexagonal patterns. The number of large particles decreased with time, so that they sparsely rimmed the gap junction in postnatal animals.
This suggests that large particles are precursors of typical gap junctional particles, and that they participate in rapid growth of the gap junction during late embryonic development. It may be also possible that large particles represent functionally different gap junctions.
Similar content being viewed by others
References
Albertini DF, Anderson E (1975) Structural modifications of lutein cell gap junctions during pregnancy in the rat and the mouse. Anat Rec 181:171–194
Decker RS (1976) Hormonal regulation of gap junction differentiation. J Cell Biol 69:669–685
Decker RS, Friend DS (1974) Assembly of gap junctions during amphibian neurulation. J Cell Biol 62:32–47
Ermak TH, Rothman SS (1984) Increase in zymogen granule volume accounts for increase in volume density during prenatal development of pancreas. Anat Rec 207:487–501
Friend DS, Gilula NB (1972) Variations in tight and gap junctions in mammalian tissues. J Cell Biol 53:758–776
Ginzberg RD, Gilula NB (1979) Modulation of cell junctions during differentiation of the chicken otocyst sensory epithelium. Dev Biol 68:110–129
Goodenough DA, Gilula NB (1974) The splitting of hepatocyte gap junctions and zonulae occludentes with hypertonic disaccharides. J Cell Biol 61:575–590
Johnson R, Hammer M, Sheridan J, Revel J-P (1974) Gap junction formation between reaggregated Novikoff hepatoma cells. Proc Natl Acad Sci USA 71:4536–4540
Loewenstein WR (1979) Junctional intercellular communication and the control of growth. Biochem Biophys Acta 560:1–65
Meyer R, Posalaky Z, McGinley D (1977) Intercellular junction development in maturing rat seminiferous tubules. J Ultrastruct Res 61:271–283
Montesano R, Philippeaux M-M (1981) Membrane particle arrays in SV 40-transformed 3T3 cells. J Cell Sci 47:311–330
Parsa I, Marsh WH, Fitzgerald PJ (1969) Pancreas acinar cell differentiation. I. Morphologic and enzymatic comparisons of embryonic rat pancreas and pancreatic anlage grown in organ culture. Am J Pathol 57:457–521
Pictet R, Rutter WJ (1972) Development of the embryonic endocrine pancreas. In: Handbook of physiology. Sec 7. Vol 1. American Physiological Society, Washington, DC, pp 25–56
Pictet RL, Clark WR, Williams RH, Rutter WJ (1972) An ultrastructural analysis of the developing embryonic pancreas. Dev Biol 29:436–467
Revel J-P, Yip P, Chang LL (1973) Cell junctions in the early chick embryo — A freeze etch study. Dev Biol 35:302–317
Rutter WJ, Ball WD, Bradshow WS, Clark WR, Sanders TG (1967) Morphological and molecular analogy in cytodifferentiation. In: Schneyer LH, Schneyer CA (eds) Secretory mechanisms of salivary glands. Academic press, New York, pp 238–253
Rutter WJ, Kemp JD, Bradshaw WS, Clark WR, Ronzio RA, Sanders TG (1968) Regulation of specific protein synthesis in cytodifferentiation. J Cell Physiol 72 Suppl 1:1–18
Sheridan JD (1976) Cell coupling and cell communication during embryogenesis. In: Poste G, Nicolson GL (eds) The cell surface in animal enbryogesisis and development. Elsevier/North-Holland Biomedical Press, Amsterdam, pp 409–447
Spooner BS, Walther BT, Rutter WJ (1970) The development of the dorsal and ventral mammalian pancreas in vivo and in vitro. J Cell Biol 47:235–246
Staehelin LA (1972) Three types of gap junctions interconnecting intestinal epithelial cells visualized by freeze-etching. Proc Natl Acad Sci USA 69:1318–1321
Staehelin LA (1974) Structure and function of intercellular junctions. Int Rev Cytol 39:191–283
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yamamoto, M., Kataoka, K. Large particles associated with gap junctions of pancreatic exocrine cells during embryonic and neonatal development. Anat Embryol 171, 305–310 (1985). https://doi.org/10.1007/BF00347019
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00347019