Summary
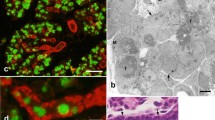
In human submandibular glands, two types of myoepithelial cells can be distinguished in serial, ultrathin sections. The dark myoepithelial cell type was stellate in shape and exhibited a pronouneced electron density due to numerous myofilaments with focal densities. Dark cell types accounted for the greater part (76%) of the myoepithelial cells and furthermore showed adenosine triphosphatase activity. This type of myoepithelial cell is considered to be that previously observed in mammalian salivary glands. Occasionally, desmosomes could be found between the processes of adjacent dark myoepithelial cell types, which is appropriate with respect to the strong compression of acinar or intercalated duct cells. The light myoepithelial cell type was large and ellipsoid with a few short-thick processes, and was characterized by an electron lucent cytoplasm which included scant and unevenly distributed myofilaments. Light cell types showed positive adenosine triphosphatase activity and accounted for only a small part (17%) of the myoepithelial cell number. Transitional forms between these two types were also observed. The light myoepithelial cell type may mature into the dark myoepithelial cell type by means of the transitional form. In addition, clear cells were sometimes encountered between the myoepithelial cell and the acinar or intercalated duct cells.
Similar content being viewed by others
References
Auger DW, Harrison JD (1982) Ultrastructural phosphatase cytochemistry of the intercalary ducts of the parotid and submandibular salivary glands of man. Arch Oral Biol 27:79–81
Batsakis JG, Kraemer B, Sciubba JJ (1983) The pathology of head and neck tumors: The myoepithelial cell and its participation in salivary gland neoplasia, part 17. Head Neck Surg 5:222–233
Cowley LH, Shackleford JM (1970) An ultrastructural study of the submandibular glands of the squirrel monkey, Saimiri sciureus. J Morphol 132:117–136
Cutler LS, Chaudhry A, Innes DJ (1977) Ultrastructure of the parotid duct. Cytochemical studies of the striated duct and papillary cystadenoma lymphomatosum of the human parotid gland. Arch Pathol Lab Med 101:420–424
Franke WW, Schmid E, Freudenstein C, Appelhans B, Osborn M, Weber K, Keenan TW (1980) Intermediate-sized filaments of the prekeratin type in myoepithelial cells. J Cell Biol 84:633–654
Garrett JR, Emmelin N (1979) Activities of salivary myoepithelial cells: A review. Med Biol 57:1–28
Garrett JR, Harrison JD (1970) Alkaline-phosphatase and adenosine-triphosphatase histochemical reactions in the salivary glands of cat, dog and man, with particular reference to the myoepithelial cells. Histochemie 24:214–229
Harrison JD (1974) Minor salivary glands of man: enzyme and mucosubstance histochemical studies. Histochem J 6:633–647
Innes DJ, Cutler LS (1978) Phosphatase enzymes. Cytochemical study of pleomorphic adenoma and normal human salivary glands. Arch Pathol Lab Med 102:90–94
Kawabata I, Kurosumi K (1976) Transmission and scanning electron microscopy of the human ceruminous apocrine gland II. Myoepithelial cells. Arch Histol Jpn 39:231–255
Leeson TS, Leeson CR (1971) Myoepithelial cells in the exorbital lacrimal and parotid glands of the rat in frozen-etched replicas. Am J Anat 132:133–146
Lojda Z, Gossrau R, Schiebler TH (1979) Enzyme histochemistry. A laboratory manual. Springer, Berlin Heidelberg New York
Pinkstaff CA (1980) The cytology of salivary glands. Int Rev Cytol 63:141–261
Radnor CJP (1972) Myoepithelial cell differentiation in rat mammary glands. J Anat 111:381–398
Shimono M, Yamamura T, Fumagalli G (1980) Intercellular junctions in salivary glands: Freeze-fracture and tracer studies of normal rat sublingual gland. J Ultrastruct Res 72:286–299
Smith CA, Monaghan P, Munro Neville A (1984) Basal clear cells of the normal human breast. Virchows Arch [A] 402:319–329
Stirling JW, Chandler JA (1976) The fine structure of the normal, resting terminal ductal-lobular unit of the female breast. Virchows Arch [A] 372:205–226
Strum JM (1978) Estrogen-induced alterations in the myoepithelial cells of the rat mammary gland. Cell Tissue Res 193:155–161
Takahashi N (1958) Electron microscopic studies on the ectodermal secretory glands in man. II. The fine structures of the myoepithelium in the human mammary and salivary glands. Bull Tokyo Med Dent Univ 5:177–192
Tamarin A (1966) Myoepithelium of the rat submaxillary gland. J Ultrastruct Res 16:320–338
Tandler B (1965) Ultrastructure of the human submaxillary gland. III. Myoepithelium. Z Zellforsch 68:852–863
Tandler B, Denning CR, Mandel ID, Kutscher AH (1970) Ultrastructure of human labial salivary glands. III. Myoepithelium and ducts. J Morphol 130:227–246
Taugner R, Schiller A (1980) Gap junctions on myoepithelial cells. Cell Tissue Res 206:65–72
Yoshihara T (1984a) Ultracytochemical localization of ouabain-sensitive, K+-paranitrophenyl phosphatase, alkaline phosphatase and Mg2+-adenosine triphosphatase activities in the salivary glands. Nippon Jibiinkoka Gakkai Kaiho 87:481–488 (Japanese)
Yoshihara T (1984b) Ultracytochemical studies on the filaments of myoepithelial cell in the human salivary gland. Nippon Jibiinkoka Gakkai Kaiho 87:714–720 (Japanese)
Young JA, van Lennep EW (1978) The morphology of salivary glands. Academic Press, London New York San Francisco
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nagashima, Y., Ono, K. Myoepithelial cell ultrastructure in the submandibular gland of man. Anat Embryol 171, 259–265 (1985). https://doi.org/10.1007/BF00347014
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00347014