Summary
Cotton and maize plants were grown under full sunlight in glass houses containing normal ambient partial pressure of CO2 (330±20 μbar) and enriched partial pressure of CO2 (640 ±15 μbar) with four levels of nitrogen nutrient. In 40 day old cotton plants grown in high CO2, there was a 2-fold increase in day weight and a 1.6-fold increase in leaf area compared with plants grown in ambient CO2. In 30 day old maize plants there was only 20% increase in dry weight in plants grown in 640 μbar CO2 compared with plants grown in 330 μbar and no significant increase in leaf area. In both species, at both CO2 treatments, dry weight and leaf area decreased in similar proportion with decreased nitrogen nutrient.
The increase of leaf area in cotton plants at high CO2 caused a reduction of total nitrogen on a dry weight basis. In cotton assimilation rate increased 1.5 fold when plants were grown with high nitrogen and high CO2. The increase was less at lower levels of nitrate nutrient. There was a 1.2 fold increase in assimilation rate in maize grown at high CO2 with high nitrate nutrient.
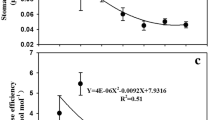
Cotton and maize grown in high CO2 had a lower assimilation rate in ambient CO2 compared to plants grown in normal ambient air. This difference was due to the reduction in RuBP carboxylase activity. Water use efficiency was doubled in both cotton and maize plants grown at high CO2 in all nutrient treatments. However, this increase in water use efficiency was due primarily to reduced transpiration in some treatments and to increased assimilation in others. These data show that plant responses to elevated atmospheric partial pressure of CO2 depend on complex of partially compensatory processes which are not readily predictable.
Similar content being viewed by others
References
Arnon, D.I.: Copper enzyme in isolate chloroplasts: polyphenol-oxidase in Beta vulgaris. Plant Physiol. 24, 1–15 (1949)
Baes, C.F., Goeller, H.E., Olson, J.C., Rotty, R.M.: Carbon dioxide and climate: The uncontrolled experiment. Am. Scientist 65, 310–320 (1977)
Bishop, P.M., Whittingham, C.P.: The photosynthesis of tomato plants in a carbon dioxide enriched atmosphere. Photosynthetica 2, 31–38 (1968)
Cooper, R.L., Brun, W.A.: Response of soybeans to a carbon dioxide-enriched atmosphere. Crop Sci. 7, 455–457 (1967)
Davidson, R.L.: Effects of soil nutrients and moisture on root/shoot ratio in Lolium perenne L. and Trifolium repens L. Ann. Bot. 33, 571–577 (1969)
Farquhar, G.D., Dubbe, D.R., Raschke, K.: Gain of the feedback loop involving carbon dioxide and stomata: Theory and measurement. Plant Physiol. 62, 406–412 (1978)
Ford, M.A., Thorne, G.N.: Effect of CO2 concentration on growth of sugar-beet, barley, kale and maize. Ann. Bot. 31, 639–644 (1967)
Frydrych, J.: Photosynthetic characteristics of cucumber seedlings grown under two levels of carbon dioxide. Photosynthetica 10, 335–338 (1976)
Gaastra, P.: Photosynthesis of crop plants as influence by light carbon dioxide, temperature and stomatal diffusion resistance. Meded. Landbouwhogesch. Wageningen 59, 1–68 (1959)
Gifford, R.M.: Growth pattern, carbon dioxide exchange and dry weight distribution in wheat growing under differing photosynthetic environments. Aust. J. Plant Physiol. 4, 99–100 (1977)
Hatch, M.D., Oliver, I.R.: activation and inactivation of phosphoenolpyruvate carboxylase in leaf extracts from C4 species. Aust. J. Plant Physiol. 5, 571–580 (1978)
Hewitt, E.J., Smith, T.A.: Plant mineral nutrition. The English University Press, London, 1975
Hofstra, G., Hesketh, J.D.: The effects of temperature and CO2 enrichment on photosynthesis in soybean. In: Environmental and biological control of photosynthesis (R. Marcelle, ed.) pp. 71–80. The Hagua, Junk 1975
Imai, K., Murata, Y.: Effect of carbon dioxide concentration on growth and dry matter production of crop plants. I. Effects on leaf area, dry matter, tillering, dry matter distribution ratio and transpiration. Proc. Crop Sci. Japan 45, 598–606 (1976)
Imai, K., Murata, Y.: Effect of carbon dioxide concentration on growth and dry matter production of crop plants. II. Specific and varietal differences in response of dry matter production. Japan J. Crop Sci. 46, 291–297 (1977)
Imai, K., Murata, Y.: Effect of carbon dioxide concentration on growth and dry matter production of crop plants. III. Relationship between CO2 concentration and nitrogen nutrition in some C3- and C4-species. Japan J. Crop Sci. 47, 118–123 (1978)
Imai, K., Murata, Y.: Effect of carbon dioxide concentration on growth and dry matter production of crop plants. IV. After-effects of carbon dioxide treatments on the apparent photosynthesis, dark respiration and dry matter production. Japan J. Crop Sci. 47, 330–335, 1978a
Lorimer, G.H., Badger, M.R., Andrews, T.J.: D-Ribulose-1,5-bisphosphate carboxylase-oxygenase: Improved methods for the activation and assay of catalytic activities. Anal. Biochem. 78, 66–75 (1977)
Lowry, O.M., Rosebrough, N.T., Farr, A.L., Randall, J.R.: Protein measurement with folic phenol reagent. J. Biol. Chem. 193, 263–275 (1951)
Neales, T.F., Nicholls, A.O.: Growth responses of young wheat plants to a range of ambient CO2 levels. Aust. J. Plant Physiol. 5, 45–59 (1978)
Raper, C.D., Jr., Peedin, G.F.: Photosynthetic rate during steady-state growth as influenced by carbon-dioxide concentration. Bot. Gaz. 139, 147–149 (1978)
Raschke, K.: Stomatal action. Ann. Rev. Plant Physiol. 26, 309–339 (1975)
Tognoni, F., Halevy, A.H., Wittwer, S.H.: Growth of bean and tomato plants as affected by root absorbed growth substances and atmospheric carbon dioxide. Planta (Berl.) 72, 43–52 (1967)
Wong, S.C., Cowan, I.R., Farquhar, G.D.: Leaf conductance in relation to assimilation in Eucalyptus pauciflora Sieb. cx Spreng: Influence of irradiance and partial pressure of carbon dioxide. Plant Physiol. 62, 670–674 (1978)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wong, S.C. Elevated atmospheric partial pressure of CO2 and plant growth. Oecologia 44, 68–74 (1979). https://doi.org/10.1007/BF00346400
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00346400